Discuss in detail about DAS guidelines, include all the topics, sub topics and all information about it with respect to Anaesthesiology
DAS difficult airway society guidelines anaesthesiology 2015 2025 complete topics
DAS guidelines all topics subtopics airway management anaesthesia
"difficult airway society" guidelines intubation adults
DAS 2025 guidelines all sections plan A B C D preoxygenation CICO extubation obstetric paediatric
DAS guidelines complete list obstetric extubation paediatric awake tracheal intubation ICU 2024 2025
DAS (Difficult Airway Society) Guidelines in Anaesthesiology — A Comprehensive Overview
1. HISTORY AND BACKGROUND
1.1 Origins
- DAS was formed in the UK and published its first intubation guidelines in 2004, replacing earlier ad hoc approaches.
- Despite their dissemination, NAP4 (2011) documented that serious airway complications — including death and brain damage — continued to occur.
- The 2015 guidelines responded to NAP4 findings with a major emphasis on managing failure, human factors, and front-of-neck access (FONA).
- The 2025 guidelines represent a philosophical shift from "managing failure" to "optimising for success," driven by advances in video laryngoscopy, high-flow nasal oxygen (HFNO), and physiological understanding.
1.2 Scope of DAS Guidelines
- Unanticipated difficult tracheal intubation in adults (2015, updated 2025)
- Awake tracheal intubation (ATI) in adults (2019)
- Tracheal extubation in adults (2012)
- Obstetric difficult/failed intubation (OAA/DAS joint, 2015)
- Paediatric difficult airway management
- Tracheal intubation in critically ill adults (DAS/ICS, 2018)
- Airway management in suspected cervical spine injury (DAS/AoA/BSOA/ICS, 2024)
- Haematoma after thyroid surgery
- Tracheostomy management (National Tracheostomy Safety Project)
2. DAS 2025 GUIDELINES — UNANTICIPATED DIFFICULT TRACHEAL INTUBATION IN ADULTS
2.1 Core Philosophy: From Failure Management to Success Optimisation
- 2015 focus: What to do when things fail
- 2025 focus: How to maximise first-attempt success and maintain continuous oxygenation throughout
- Central organising principle: "Oxygenation is the mission; everything else is a tactic"
2.2 Pre-Procedure Planning and Assessment
Airway Assessment
- Every patient must have a pre-operative airway assessment addressing all four Plans (A, B, C, D)
- Assessment should incorporate history of previous difficulty
- Point-of-care ultrasound (POCUS) endorsed for airway anatomy and gastric assessment
- If any difficulty anticipated in Plans A–D → consider awake tracheal intubation
Communication and Team Briefing
- Airway strategy must be communicated with the anaesthetic assistant
- Airway management discussed during the theatre team briefing
- Equipment for Plans A–D must be immediately available wherever airway management is performed
- Out-of-hours airway management carries increased risk; teams must plan accordingly
- Optimal location for airway management must be considered
2.3 Preoxygenation
- Optimise patient position before preoxygenation (ramped position for obese patients, head elevated)
- Target: FiO₂ ≥ 0.9; ETO₂ ≥ 90% before induction where feasible
- High-flow nasal oxygen (HFNO) / transnasal humidified rapid-insufflation ventilatory exchange (THRIVE) during apnoea extends safe apnoea time
- Nasal cannula oxygen at 15 L/min maintained throughout all phases
2.4 The A–B–C–D Algorithm
PLAN A — Tracheal Intubation (Facemask Ventilation + Laryngoscopy)
- Video laryngoscopy (VL) is now the default first-line device, not the backup
- Maximum 3 + 1 attempts maintained (3 by primary operator; 4th and final only by a more experienced colleague)
- Each subsequent attempt must involve a meaningful change: different blade/device, introducer, external laryngeal manipulation, suction, removal of cricoid force, or optimised neuromuscular block
- A suboptimal first attempt is a wasted attempt — emphasis on making the first attempt the best attempt
- Continuous oxygen delivery (nasal cannula or HFNO) throughout all attempts
- If hypoxaemia occurs at any time → abandon attempt, prioritise oxygenation with facemask ventilation
- Maintain anaesthesia between attempts
- Adequate neuromuscular blockade facilitates laryngoscopy
- Use waveform capnography to confirm tracheal placement
- Declare "failed intubation" clearly before moving to Plan B
- Optimise head and neck position (sniffing/ramped)
- Preoxygenate
- Adequate neuromuscular blockade
- Direct or video laryngoscopy (max 3+1 attempts)
- External laryngeal manipulation
- Bougie use
- Remove cricoid pressure if applied and unhelpful
- Maintain oxygenation and anaesthesia throughout
PLAN B — Supraglottic Airway Device (SAD) Insertion: Maintaining Oxygenation
- Plan B is no longer merely a "rescue pause" — it reframes the SAD as a strategic oxygenation platform
- Some patients cannot be woken up (critically ill, physiologically complex) — Plan B must account for this
- Second-generation SADs preferred (e.g., i-gel, LMA Supreme, ProSeal) — they provide a better seal and have gastric drain ports
- Maximum 3 attempts at SAD insertion (change device or size between attempts)
- If SAD succeeds → "Stop and Think" — four options:
- Wake the patient up (preferred if feasible)
- Intubate trachea via the SAD (fibreoptic-assisted)
- Proceed with surgery without tracheal intubation (under SAD alone)
- Surgical airway (tracheostomy or cricothyroidotomy)
- Failed SAD ventilation → declare, move to Plan C
PLAN C — Facemask Ventilation: Buying Time
- Last-resort attempt to maintain oxygenation non-invasively
- If facemask ventilation was previously impossible, consider neuromuscular blockade to enable it
- Two-person technique with jaw thrust and adjuncts (oropharyngeal/nasopharyngeal airway)
- If facemask ventilation succeeds → wake the patient up
- Failed facemask ventilation → declare CICO and proceed immediately to Plan D
PLAN D — Emergency Front-of-Neck Access (eFONA): CICO Rescue
- Declare CICO loudly — activates team response, prevents further airway instrument attempts
- One single recommended technique: Scalpel–Bougie–Tube (vertical eFONA)
- Every anaesthetist should learn and regularly train in scalpel cricothyroidotomy
- "Priming" concept (2025): the eFONA kit should be opened and ready in parallel with Plan B/C, not as an afterthought
- Stand on patient's left side (if right-handed)
- Maximum neck extension; identify laryngeal anatomy and midline with non-dominant hand
- Apply tension to skin; stabilise larynx
- Horizontal stab incision through cricothyroid membrane (CTM) with scalpel (dominant hand)
- Keep scalpel perpendicular to skin; rotate 90° so sharp edge points caudally
- Swap hands; hold scalpel with left hand, maintain traction
- Slide bougie's coudé tip down the scalpel blade into the trachea (right angle to trachea)
- Rotate and align bougie with trachea; advance 10–15 cm
- Remove scalpel; stabilise trachea
- Railroad lubricated size 6.0-mm cuffed tracheal tube over bougie; rotate as advanced
- Remove bougie; inflate cuff; confirm ventilation with waveform capnography
- High failure rates in real-world practice
- Risk of misplacement, kinking, barotrauma
- Scalpel technique is more reliable and teachable
- Percutaneous dilatational kits remain an option for experienced operators
2.5 Physiologically Difficult Airway (New in 2025)
- Patients who are hypoxaemic, acidotic, haemodynamically unstable, or in metabolic crisis
- These patients have reduced reserve and greater risk of peri-intubation cardiac arrest
- Preparation must include:
- Optimisation of haemodynamics before intubation (fluids, vasopressors)
- Preoxygenation with HFNO
- Post-intubation stabilisation planning
- Avoiding prolonged apnoea
2.6 Post-Intubation Care and Follow-Up
- Formulate an immediate post-procedure airway management plan
- Monitor for complications (oedema, trauma, haemorrhage)
- Complete airway alert form
- Explain difficulties to patient in person and in writing
- Send written report to primary care provider and local database
- Document in medical records — WHO checklist sign-out section
- Patient follow-up by anaesthetist to identify complications (airway trauma is closely linked to difficult intubation)
- All failed intubations and eFONA events should be reviewed at morbidity and mortality meetings
2.7 Human Factors and Team Performance (2015 and 2025)
- Cognitive overload under crisis degrades performance
- Use structured communication tools such as PACE (Probe, Alert, Challenge, Emergency) to communicate concerns in hierarchical environments
- Clear declaration of failure at each step prevents cognitive fixation (tunnel vision)
- Situational awareness, role assignment, and briefings are mandatory elements
- Debriefing after difficult airway events
- Human factors training should be included in simulation curricula
- Individual, team, and organisational levels all considered
2.8 Continuous Oxygenation as Central Principle (2025)
- Oxygen delivery must never stop — not between attempts, not during pauses or transitions
- Nasal cannula at ≥15 L/min as minimum; HFNO preferred during pre-oxygenation and apnoea
- This is the most significant conceptual departure from 2015
2.9 Training and Education Recommendations
- All anaesthetists must have regular training in scalpel cricothyroidotomy
- Video laryngoscopy skills must be maintained
- Institutions should support clinicians training with new equipment or guidelines
- Airway leads should reinforce familiarity with Plans A–D among all airway managers and assistants
- Simulation is the cornerstone of competency maintenance
- All individuals involved in airway management should be trained in waveform capnography interpretation
- Evidence of airway training supports annual appraisal/revalidation
3. DAS GUIDELINES FOR AWAKE TRACHEAL INTUBATION (ATI) — 2019
3.1 Indications for ATI
- Anticipated difficult airway where loss of airway on induction would be hazardous
- Full stomach with predicted difficult mask ventilation (cannot rely on rapid sequence induction rescue)
- Unstable cervical spine
- Severe obstructive sleep apnoea with anticipated difficult airway
- Upper airway pathology (tumours, abscesses, foreign bodies, angioedema)
- Morbid obesity with multiple predictors of difficulty
3.2 Contraindications
- Uncooperative patient
- Local anaesthetic allergy
- Severe respiratory distress requiring immediate intubation
- Relative: upper airway bleeding obscuring endoscopy
3.3 Technique
- Informed consent and explanation
- Glycopyrrolate (antisialagogue) to dry secretions
- Supplemental oxygen throughout
- Nose: Cocaine 4–10% or lidocaine with vasoconstrictor (shrinks mucosa, topical LA)
- Oropharynx/tongue base: Lidocaine spray 10%, gargle, or mucosal atomisation device (MAD)
- Larynx and trachea:
- Nebulised lidocaine 4%
- Transtracheal injection: lidocaine 2–4 mL through CTM
- Superior laryngeal nerve block (bilateral): lidocaine 2% lateral to thyrohyoid membrane
- Maximum safe dose: lidocaine 9 mg/kg with adrenaline (4 mg/kg without, but topical LA is poorly absorbed — higher doses are used clinically with care)
- Dexmedetomidine (most evidence) — provides sedation, anxiolysis, analgesia without respiratory depression
- Remifentanil TCI — short-acting; requires careful titration
- Midazolam — anxiolysis but risk of over-sedation; use cautiously
- Ketamine — dissociative, maintains airway reflexes; useful in anxious patients
- Avoid over-sedation — patient cooperation and airway reflexes must be maintained
- Nasal fibreoptic bronchoscopy (FOB) — most common; provides excellent visualisation
- Oral fibreoptic bronchoscopy — with bite block
- Video laryngoscopy (awake VL) — emerging evidence, comparable success rates
- Combined techniques (VL + FOB)
- SpO₂, ECG, NIBP
- Supplemental oxygen throughout
- Airway obstruction and respiratory pattern monitored
3.4 Confirmation and Post-ATI Care
- Confirm tracheal placement with waveform capnography before inducing general anaesthesia
- Document the technique and equipment used
4. DAS EXTUBATION GUIDELINES — 2012 (Updated)
4.1 Rationale
4.2 Risk Stratification
- "Low risk" — routine extubation, no specific concerns
- "At risk" — where complications may be difficult to manage; requires a structured plan
- Difficult intubation at induction
- Airway oedema (prolonged surgery, prone positioning, massive fluid resuscitation)
- Obesity
- Restricted neck mobility
- Upper airway surgery
- Shared airway cases
- Limited airway access post-surgery
4.3 Extubation Strategy
- Consider extubation at the time of intubation, not as an afterthought
- Is awake extubation safer? Is deep extubation feasible?
- Full reversal of neuromuscular blockade (confirmed with nerve stimulator or quantitative monitoring)
- Haemodynamic stability
- Adequate analgesia and antiemesis
- Patient fully conscious (awake extubation preferred in at-risk patients)
- Equipment immediately available for re-intubation
- Consider airway exchange catheter (AEC) through the tracheal tube before extubation in "at-risk" patients — allows oxygenation and serves as a guide for re-intubation if needed
- Remifentanil infusion can obtund responses during extubation of at-risk airways
- Monitoring in high-dependency setting for at-risk patients
- Supplemental oxygen
- Clear documentation of any extubation concerns
- Communicate to recovery nursing staff
4.4 Special Techniques
- Laryngeal mask airway (LMA) exchange: remove tracheal tube over an LMA (Bailey manoeuvre) for patients likely to obstruct or laryngospasm on extubation
- Remifentanil TCI during extubation to suppress coughing and bucking
- Nebulised adrenaline/dexamethasone for post-extubation stridor
5. OAA/DAS OBSTETRIC GUIDELINES — 2015
5.1 Why Obstetrics Requires Separate Guidelines
- Rapid desaturation (reduced FRC, increased oxygen consumption)
- Risk of aspiration (full stomach, raised intra-abdominal pressure, delayed gastric emptying)
- Foetal compromise — urgency can limit time for careful planning
- Haemodynamic instability (aortocaval compression)
- Airway oedema (pre-eclampsia, fluid overload)
- Failed intubation rate in obstetrics: ~1:300 (vs ~1:2,000 in general surgical patients)
5.2 Algorithm Overview
- Category 1 (immediate threat to life): rapid-sequence induction (RSI) is usually still pursued; failed intubation triggers CICO algorithm
- Non-Category 1/2: more time for awake intubation or alternative regional technique
- Preoxygenation: 3 min tidal volume breathing or 4 vital capacity breaths of 100% O₂
- Cricoid pressure applied at induction (controversial but still recommended in obstetric RSI)
- Thiopentone 4–5 mg/kg or propofol
- Suxamethonium 1.5 mg/kg (preserves fastest offset; still preferred in obstetric RSI for most indications)
- Rocuronium 1.2 mg/kg with sugammadex availability is an alternative
- Insert second-generation SAD (e.g., LMA Supreme)
- Decision: continue with SAD anaesthesia (if oxygenation maintained and surgery urgent) vs wake the patient up
- CICO → emergency front-of-neck access (scalpel cricothyroidotomy)
5.3 Key Differences from General DAS Guidelines
- More conservative attempt limits (maximum 2 laryngoscopy attempts recommended by some obstetric guidance)
- Cricoid pressure retained during RSI (debate ongoing)
- The foetal status influences the "wake up" decision
- Post-event: notify obstetric team, neonatology, complete incident reporting
6. DAS/ICS GUIDELINES FOR CRITICALLY ILL ADULTS — 2018
6.1 Why the ICU Is Different
- Most ICU patients have physiologically difficult airways (hypoxic, acidotic, haemodynamically compromised)
- Limited access, positioning difficulties, altered anatomy
- Higher failed intubation and cardiac arrest rates compared to operating theatre
- Less immediately available specialist help
6.2 Key Recommendations
- Pre-intubation optimisation: fluid resuscitation, vasopressors, correction of metabolic derangements
- HFNO or NIV for pre-oxygenation
- Video laryngoscopy as first-line in ICU (endorsed before the 2025 DAS update extended this to elective practice)
- RSI as default (vs inhalational induction used sometimes in theatres)
- Maximum 2 attempts before escalation (due to higher risk environment)
- Equipment: must have difficult airway trolley including eFONA kit at bedside
- Capnography mandatory for all intubations
- Team: ideally two operators present (one to intubate, one to assist)
- Post-intubation: sedation and ventilation strategy, haemodynamic monitoring
7. DAS GUIDELINES FOR CERVICAL SPINE INJURY — 2024
7.1 The Clinical Challenge
- Cervical spine immobilisation (manual inline stabilisation, MILS) worsens laryngoscopic view
- Risk of worsening neurological injury during intubation (though evidence is limited)
- Haemodynamic instability (cervical cord injury causes neurogenic shock)
- Concurrent head injury may limit awake technique
7.2 Key Recommendations
- MILS should be maintained during intubation (not cricoid force, which can move the spine)
- Video laryngoscopy preferred — provides better view with less cervical movement than direct laryngoscopy
- Awake fibreoptic intubation for predicted difficult airways with stable patients
- Avoid excessive cervical extension
- Succinylcholine is contraindicated after 72 hours of denervation injury (risk of hyperkalaemic cardiac arrest); rocuronium preferred
- All five institutions have agreed on a unified pathway
8. DAS HAEMATOMA AFTER THYROID SURGERY GUIDELINES
8.1 Clinical Scenario
8.2 Management
- Immediate wound opening at bedside (remove clips/sutures) to decompress haematoma — this is the priority, even before moving to theatre
- Haematoma decompression may partially restore airway patency
- Call for senior anaesthetic and surgical help simultaneously
- Attempt awake fibreoptic intubation if airway not immediately critical
- Be prepared for RSI or surgical airway
- Tracheostomy may be required if distortion is severe
- Transfer to theatre once airway is secured
9. TRACHEOSTOMY MANAGEMENT — NATIONAL TRACHEOSTOMY SAFETY PROJECT (NTSP)
9.1 Emergency Management of Tracheostomy
- Patients with a tracheostomy in situ who develop respiratory distress require a systematic approach
- NTSP algorithm: assess patency → suction → remove inner cannula → deflate cuff → attempt oral/nasal airway as backup
- If tracheostomy cannot be cleared → cover stoma and attempt oral ventilation
- Prepare for re-intubation via oral route or insertion of new tube into stoma
9.2 Tracheostomy Tube Types and Selection
- Cuffed vs uncuffed; fenestrated vs non-fenestrated
- Size selection based on patient tracheal diameter (ultrasound-guided if uncertain)
- Speaking valves (Passy Muir): one-way valve; requires cuff deflated; can be used once airway is patent
- Regular tube changes per institutional protocol
10. PAEDIATRIC DIFFICULT AIRWAY
- Airway anatomy differs fundamentally (proportionally large tongue, large occiput, short trachea, narrow subglottis)
- Inhalational induction preferred to maintain spontaneous ventilation
- Smaller SADs have different performance characteristics
- eFONA (cricothyrotomy) is anatomically more difficult in small children — needle cricothyrotomy or emergency tracheostomy may be preferred
Paediatric algorithm principles:
- Maintain spontaneous ventilation as long as possible
- Sevoflurane inhalational induction for cooperative and predicted difficult airway cases
- Limited attempts (maximum 2–3)
- Smaller video laryngoscopes increasingly available (e.g., C-MAC paediatric blades)
- Call for surgical/ENT help early in CICO
11. KEY DEVICES AND TECHNOLOGIES REFERENCED IN DAS GUIDELINES
| Device/Technology | Role in DAS Algorithm |
|---|---|
| Video laryngoscope (GlideScope, C-MAC, McGrath) | Plan A first-line (2025); adjunct in 2015 |
| Fibreoptic bronchoscope | AWake tracheal intubation; intubation via SAD |
| Second-generation SAD (i-gel, LMA Supreme, ProSeal) | Plan B |
| Bougie/introducers | Adjunct in Plan A; used in eFONA |
| HFNO (e.g., Optiflow) | Preoxygenation, apnoeic oxygenation throughout all plans |
| Waveform capnography | Mandatory confirmation at every step |
| Airway exchange catheter (AEC) | At-risk extubation |
| POCUS | Pre-procedure airway and gastric assessment |
| Neuromuscular monitoring | Confirm adequate blockade (Plan A); reverse for Plan C |
12. COMPARISON: DAS 2015 vs DAS 2025 — KEY DIFFERENCES
| Feature | DAS 2015 | DAS 2025 |
|---|---|---|
| Core philosophy | Managing failure | Optimising success |
| Primary laryngoscope | Direct laryngoscopy (VL as backup) | Video laryngoscopy as default |
| Continuous oxygenation | Recommended | Central mandatory principle |
| HFNO during apnoea | Not explicitly recommended | Explicitly endorsed |
| Physiologically difficult airway | Not addressed | Formally addressed |
| POCUS | Not mentioned | Endorsed for airway + gastric assessment |
| Attempt limits | 3+1 | 3+1 maintained |
| eFONA "priming" | Reactive | Proactive, parallel preparation |
| Waking patient up | Default after failed intubation | Context-dependent; not always possible |
| VL for Plan A | Alternative | Standard |
| Human factors | Introduced | Substantially expanded |
13. UNIVERSAL PRINCIPLES ACROSS ALL DAS GUIDELINES
- Declare failure clearly at the end of each plan before progressing
- Maintain oxygenation at all times — this is non-negotiable
- Limit attempts — each failed attempt increases trauma and reduces success likelihood
- Call for help early — do not persist alone in a deteriorating situation
- Waveform capnography for all intubations
- Document and communicate every difficult airway event
- Follow up patients after difficult airway events
- Train regularly — especially in eFONA, which is rarely needed but must be executed confidently
- Human factors matter as much as technical skill
DAS 2015 Algorithm (Overview and Detail)
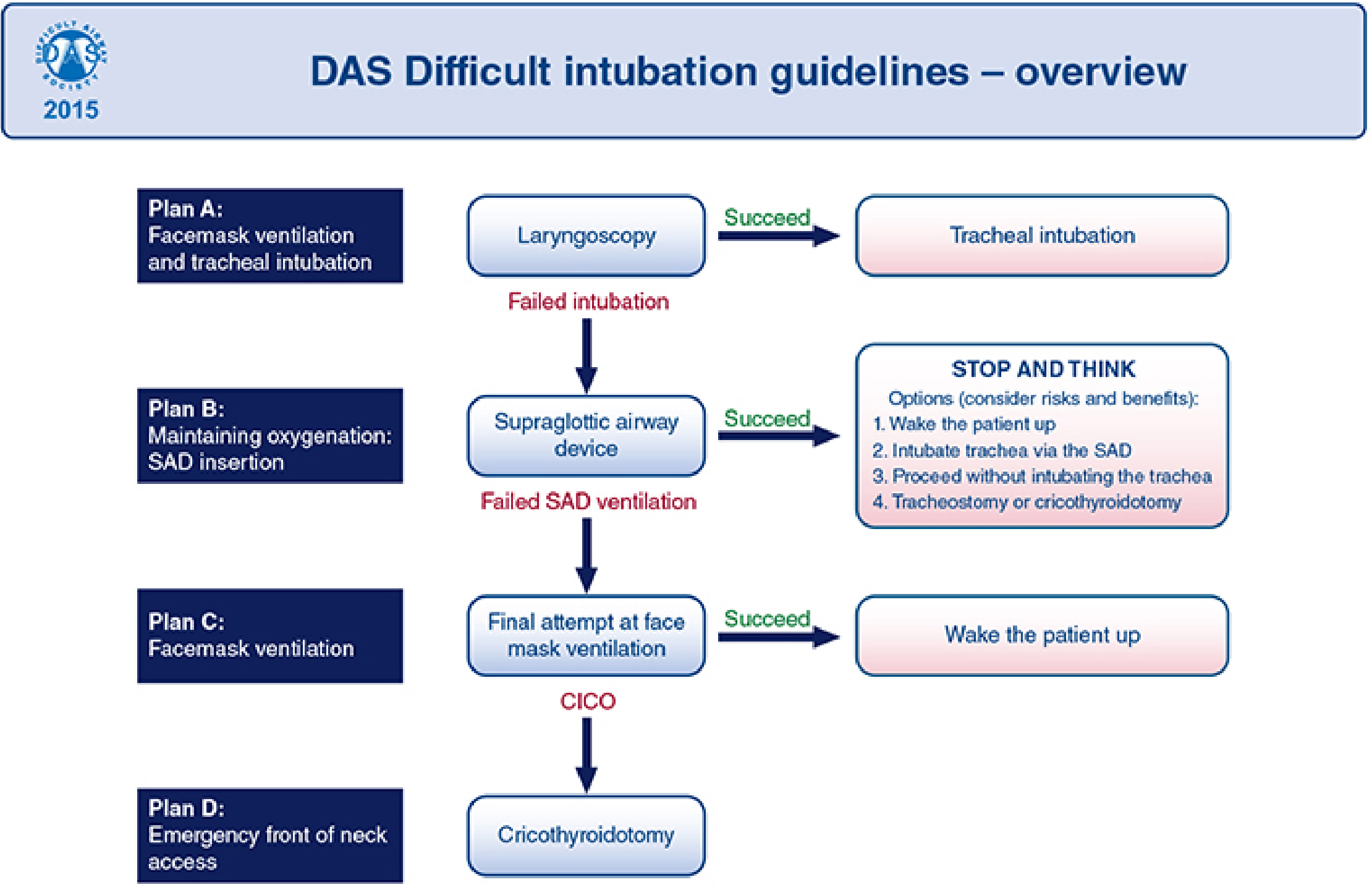
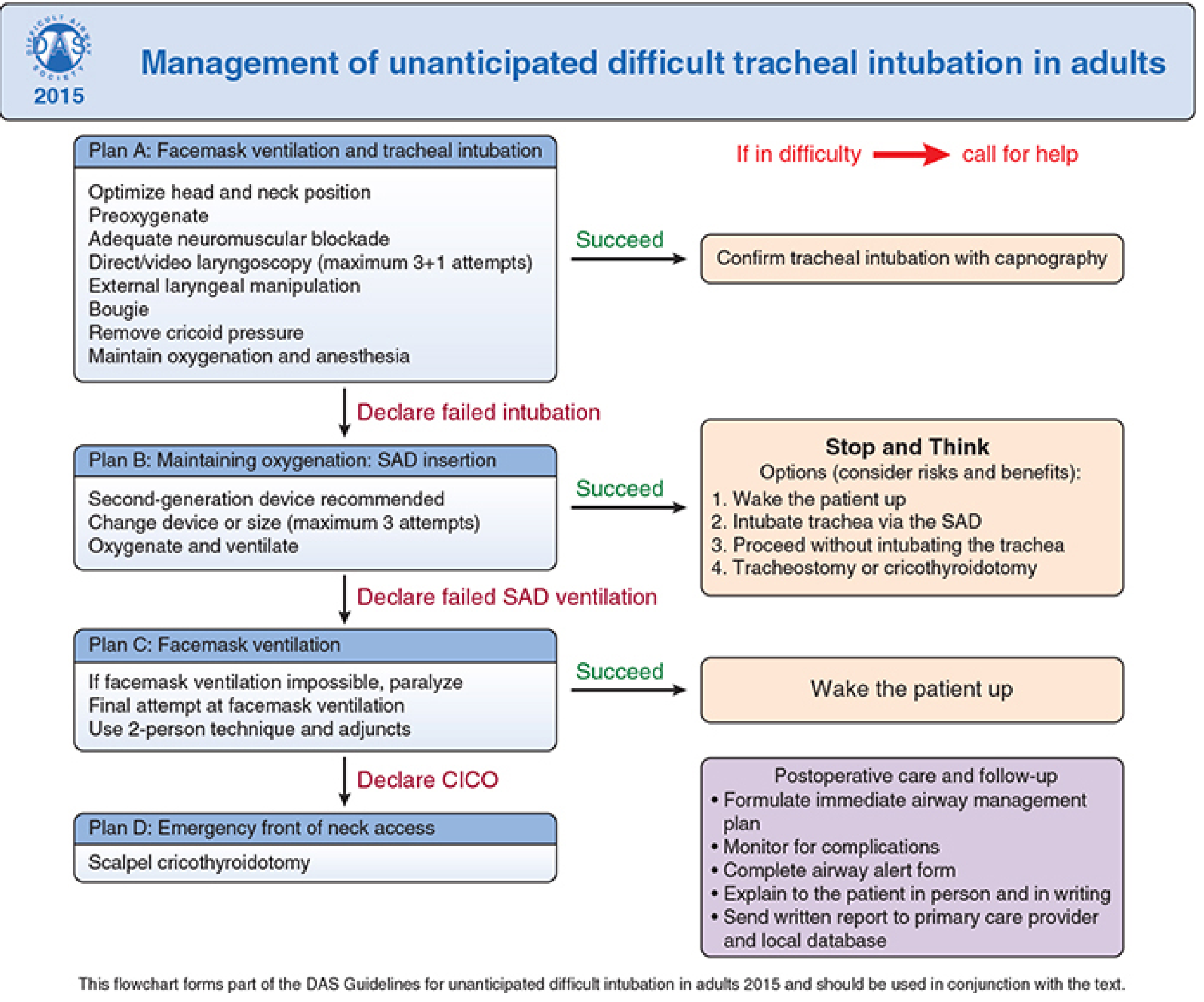
KEY REFERENCES
- Ahmad I, El-Boghdadly K, Iliff H et al. Difficult Airway Society 2025 guidelines for management of unanticipated difficult tracheal intubation in adults. Br J Anaesth. 2026 Jan. [PMID: 41203471]
- Frerk C, Mitchell VS, McNarry AF et al. Difficult Airway Society 2015 guidelines for management of unanticipated difficult intubation in adults. Br J Anaesth. 2015;115:827–848. [PMC4650961]
- Wiles MD, Iliff HA, Brooks K et al. Airway management in patients with suspected or confirmed cervical spine injury (DAS/AoA/BSOA/ICS guidelines). Anaesthesia. 2024;79:856–868. [PMID: 38699880]
- Mushambi MC, Kinsella SM et al. OAA/DAS guidelines for management of difficult and failed tracheal intubation in obstetrics. Anaesthesia. 2015.
- Higgs A, McGrath BA et al. Guidelines for management of tracheal intubation in critically ill adults (DAS/ICS). Br J Anaesth. 2018;120:323–352.
- Morgan & Mikhail's Clinical Anesthesiology, 7e — DAS Algorithm, p.608–611
- Miller's Anesthesia, 10e — Extubation and DAS risk stratification, p.5959–5961
Discuss in detail about 2025 DAS guidelines, explain all the managements
DAS 2025 guidelines unanticipated difficult tracheal intubation adults full text all recommendations
DAS 2025 all recommendations airway assessment preoxygenation plan A B C D CICO eFONA RSI physiologically difficult airway obesity human factors POCUS documentation training
DAS 2025 all recommendations plan A B C D preoxygenation RSI physiologically difficult obesity POCUS human factors documentation education training
DAS 2025 guidelines recommendations RSI physiologically difficult airway obesity POCUS peroxygenation plan A B C D eFONA complete detail
DAS 2025 POCUS human factors documentation post-intubation care training 65 recommendations detail
DAS 2025 all detailed recommendations plan A B C D RSI physiologically difficult airway POCUS drugs neuromuscular blockade monitoring post-intubation
DAS 2025 monitoring drugs neuromuscular blockade capnography post-intubation care recommendations detail anaesthesia
DAS 2025 Guidelines — Detailed Comprehensive Discussion
OVERVIEW AND METHODOLOGY
The 2015 guidelines focused on managing failure. The 2025 guidelines focus on maximising the likelihood of success at the first attempt, while ensuring continuous oxygenation and clear escalation when plans fail.
SECTION 1: AIRWAY ASSESSMENT
Rationale
What to Assess
- Clinical history — prior anaesthetic records, previous difficult intubation, previous surgery to the head/neck/spine, OSA, snoring, stridor
- Physical examination with bedside tests — mouth opening (inter-incisor distance), Mallampati score, thyromental distance, neck mobility, neck circumference, teeth (loose, prominent, crowded), jaw protrusion
- Relevant investigations — CT/MRI of the airway when pathology is suspected; nasoendoscopy for suspected upper airway tumours or obstruction
- The physiologically difficult airway must also be considered (see Section 10)
Scoring Systems
- HEAVEN criteria — particularly useful in pre-hospital care (Hypoxia, Extremes of size, Anatomical abnormality, Vomit/blood/fluid, Exsanguination/poor vascular access, Neck mobility)
- MACOCHA score — validated in critical care for predicting difficult intubation in ICU
Recommendations
- Airway assessment must be performed before induction of anaesthesia
- Assessment should include history, examination with bedside tests, and when appropriate, review of relevant investigations
- The physiologically difficult airway should also be considered
SECTION 2: PLANNING AND STRATEGY
Airway Management Strategy
Key Elements
- Strategy should be guided by any history of previous difficulty
- If difficulty is anticipated in Plans A, B, C, or D → consider awake tracheal intubation
- Strategy must be clearly communicated to the anaesthetic assistant before starting
- Airway management should be discussed during the theatre team briefing (pre-list WHO safety checklist)
- Equipment for Plans A–D must be immediately available in all locations where airway management is performed
- Out-of-hours airway management carries increased risk — teams must plan accordingly and senior help should be more readily available
- Optimal location for airway management should be considered (e.g., operating theatre vs ICU vs ward)
- Checklists are useful during emergency tracheal intubation to improve adherence and optimise teamworking
Recommendations
- Airway management strategy must address anticipated difficulty in: A) tracheal intubation; B) SAD insertion/ventilation; C) facemask ventilation; D) emergency front-of-neck airway
- Strategy should be guided by history of previous difficulty
- If any difficulty is anticipated in Plans A–D, awake tracheal intubation should be considered
- Strategy must be clearly communicated to the anaesthetic assistant
- Airway management discussed as part of the theatre team briefing
- Patient position should be optimised before preoxygenation and for tracheal intubation
- Equipment for Plans A–D must be immediately available in all areas
- Out-of-hours risks must be planned for
- Optimal location for airway management should be considered
- Consider a checklist when performing emergency tracheal intubation
SECTION 3: MONITORING
Minimum Monitoring Standards
- Inspired and expired oxygen monitoring
- Waveform capnography
Waveform Capnography — The Gold Standard
- Must be checked before induction (confirms circuit function)
- Audible SpO₂ tones must be enabled before induction — provides real-time audio feedback without needing to watch the monitor
- Continuous, uninterrupted waveform capnography must be used throughout all phases of airway management
- Confirms tracheal placement; absence of waveform despite intubation = oesophageal intubation until proven otherwise
- Used to confirm ventilation at every step of the algorithm
Neuromuscular Monitoring
- Quantitative neuromuscular monitoring (acceleromyography or kinemyography) should be used to confirm adequate neuromuscular block before tracheal intubation attempts (where feasible)
- Guides timing of intubation attempts (ensures full paralysis at the moment of laryngoscopy)
- Reduces complications during maintenance and risks at extubation
- Institutions should provide equipment enabling patient monitoring per current Association of Anaesthetists recommendations
Recommendations
- Waveform capnography checked before induction
- Audible SpO₂ tones enabled before induction
- Continuous, uninterrupted waveform capnography throughout all phases of airway management
- Quantitative neuromuscular monitoring to confirm adequate block before intubation attempts, where feasible
- Institutions to provide appropriate monitoring equipment
SECTION 4: DRUGS
Neuromuscular Blocking Agents (NMBAs)
- NMBAs should be routinely used to facilitate tracheal intubation — this is a strengthened recommendation compared to 2015, where their use was conditional
- Rationale: adequate neuromuscular blockade significantly improves laryngoscopic view, reduces complications (airway trauma), and increases first-pass success
- Waiting for full onset of neuromuscular blockade before attempting laryngoscopy is essential
- Choice of agent:
- Suxamethonium (succinylcholine) 1–1.5 mg/kg — fastest onset and offset; preferred for RSI when rapid return of spontaneous ventilation is desired
- Rocuronium 1.2 mg/kg — equivalent intubating conditions in 60–90 seconds; preferred when suxamethonium is contraindicated; sugammadex (16 mg/kg) available for immediate reversal if CICO
- The availability of sugammadex changes the risk-benefit calculation for using rocuronium in anticipated difficult airways
Recommendation
- Neuromuscular blocking agents should be routinely used to facilitate tracheal intubation
SECTION 5: PEROXYGENATION
Definition (New Unified Term — 2025)
- Pre-oxygenation — before induction
- Apnoeic oxygenation — during apnoea/intubation attempts
- Continuous oxygenation during laryngoscopy — nasal cannula O₂ maintained while the scope is in the mouth
Pre-oxygenation
- All patients must be pre-oxygenated before induction of general anaesthesia
- Performed in the head-up position (≥20°) — reduces atelectasis, increases FRC, prolongs safe apnoea time
- Technique must allow positive pressure, where feasible — use a well-fitting facemask with PEEP valve or tight-fitting anaesthetic mask on the breathing circuit
- Target: end-tidal O₂ ≥ 90% (= FiO₂ replacement of nitrogen)
High-Flow Nasal Oxygen (HFNO) for Peroxygenation
- In patients at risk of difficult airway management, peroxygenation should be used, ideally with HFNO (e.g., Optiflow at 40–70 L/min, FiO₂ 1.0)
- HFNO provides apnoeic oxygenation during intubation attempts — extends safe apnoea time significantly (demonstrated to extend safe apnoea beyond 10 minutes in some patients)
- Nasal cannula at ≥ 15 L/min O₂ as minimum during all intubation attempts
Recommendations
- All patients should be pre-oxygenated before induction
- Pre-oxygenation in head-up position with positive pressure technique, where feasible
- In patients at risk of difficult airway, peroxygenation should be used, ideally with HFNO
SECTION 6: PLAN A — TRACHEAL INTUBATION
Core Principle
Patient Positioning
- Optimal position before preoxygenation and intubation
- Sniffing position (head extended, neck flexed) for standard patients — aligns oral, pharyngeal, and laryngeal axes
- Ramped position for obese patients (ear-to-sternal notch level)
Video Laryngoscopy — Now First-Line
- Video laryngoscopy (VL) is the recommended first-line device for Plan A (major change from 2015)
- Evidence: VL consistently improves laryngoscopic view grade and increases first-attempt success
- VL does not guarantee successful intubation — tube delivery can be difficult despite improved view; use adjuncts (stylet, bougie, angulated blade)
- Direct laryngoscopy remains acceptable but VL should be default where available
Attempt Limits: 3 + 1 Rule (Maintained from 2015)
- Maximum 3 attempts by the primary operator
- A 4th and final attempt only by a more experienced colleague
- Each subsequent attempt must involve a meaningful change to increase likelihood of success:
- Change laryngoscope type or blade size
- Use or change an introducer (bougie/stylet)
- Change operator
- External laryngeal manipulation (ELM) or airway suction
- Remove cricoid force if applied
- Optimise neuromuscular blockade
- Each failed attempt increases airway trauma and reduces the chance of success → the "death by a thousand laryngoscopies" phenomenon
During Intubation Attempts
- Maintain continuous nasal O₂ (nasal cannula or HFNO)
- Maintain anaesthesia between attempts
- If hypoxaemia occurs at any time → abandon the attempt immediately, prioritise oxygenation with facemask ventilation
- Continue facemask ventilation between attempts
- Waveform capnography mandatory to confirm tube placement
- Declare "failed intubation" clearly and unambiguously before moving to Plan B
Plan A Algorithm Flow
Optimise position → Pre-oxygenate (HFNO) → Adequate NMB
→ Video laryngoscopy (max 3+1 attempts, meaningful change each time)
→ Capnography confirmation
→ SUCCESS: post-intubation care
→ FAILURE: Declare failed intubation → Plan B
SECTION 7: PLAN B — SUPRAGLOTTIC AIRWAY DEVICE (SAD)
Core Principle
Key Change from 2015
Device Selection
- Second-generation SADs are recommended (e.g., i-gel, LMA Supreme, LMA ProSeal)
- Advantages of second-generation devices: higher oropharyngeal seal pressure, integral gastric drain channel (reduces aspiration risk), better performance in difficult airways
- Evidence supports second-generation devices over classic LMA in failed intubation scenarios
Attempt Limits
- Maximum 3 attempts at SAD insertion
- Between attempts: change device size or type
- Ensure facemask ventilation continues between SAD insertion attempts
"Stop and Think" After Successful SAD
| Option | When to Use |
|---|---|
| 1. Wake the patient up | Safest option if surgery can be postponed; patient not physiologically compromised |
| 2. Intubate trachea via SAD | Using fibreoptic bronchoscope through SAD; when tracheal intubation is essential and patient cannot be woken |
| 3. Proceed with SAD as sole airway | When surgery is urgent, short, low-risk for aspiration, and SAD provides adequate ventilation |
| 4. Surgical airway | If all other options are not feasible or high-risk |
Priming for eFONA (New 2025 Concept)
- Confirm that the eFONA kit is immediately accessible
- Declare clearly who is the most skilled individual to perform eFONA
- This preparation runs in parallel with Plan B attempts — it does not wait for Plan C or D
Transitioning
- Declared failed SAD ventilation → Plan C
- Continue oxygen delivery via nasal cannula even if SAD fails
SECTION 8: PLAN C — FINAL ATTEMPT AT FACEMASK VENTILATION
Core Principle
Technique
- If facemask ventilation was previously impossible or inadequate → consider ensuring full neuromuscular blockade (if not already achieved) to relax pharyngeal muscles
- Two-person technique: one operator performs jaw thrust with both hands on the mask (EC grip); second person compresses the bag
- Use oropharyngeal airway (Guedel), nasopharyngeal airway, or both
- Apply optimal head position and jaw thrust throughout
If Plan C Succeeds
- Wake the patient up (preferred)
- Reconsider surgical, regional, or awake intubation options
If Plan C Fails
- Declare CICO (Cannot Intubate, Cannot Oxygenate) — loudly and explicitly
- This is a life-threatening emergency
- Immediately proceed to Plan D
Clinical Note (2025)
SECTION 9: PLAN D — EMERGENCY FRONT-OF-NECK AIRWAY (eFONA)
Trigger: CICO Declaration
- CICO is declared when neither tracheal intubation nor non-invasive oxygenation (via SAD or facemask) can be achieved
- Time-critical emergency — prolonged hypoxia leads to cardiac arrest and brain damage
- All prior airway attempts must stop
- Immediately proceed to Plan D
Recommended Technique: Scalpel–Bougie–Tube (Vertical eFONA)
- Scalpel (No. 10 blade)
- Bougie (standard tracheal tube introducer)
- Cuffed size 6.0-mm tracheal tube (lubricated)
- Waveform capnography (for confirmation)
- Maintain oxygen delivery via upper airway (nasal cannula) even during eFONA
- Ensure full neuromuscular blockade (facilitates neck extension)
- Position patient in maximal neck extension
- Stand on patient's left side (if right-handed; reverse if left-handed)
- Palpate and identify laryngeal anatomy in maximum neck extension — locate the midline and cricothyroid membrane (CTM) with the non-dominant hand
- Apply skin tension with non-dominant hand; stabilise the larynx
- Make a bold horizontal stab incision through the CTM with the scalpel (dominant hand), perpendicular to the skin
- Keep scalpel perpendicular; rotate 90° so the sharp edge points caudally
- Swap hands — hold scalpel with left hand; pull laterally toward you, keeping handle vertical
- Pick up the bougie with your right hand
- Hold the bougie at a right angle to the trachea; slide the coudé tip down the far side of the scalpel blade into the trachea
- Rotate and align the bougie with the trachea; advance gently 10–15 cm
- Remove the scalpel
- Stabilise the trachea and apply skin tension with the left hand
- Railroad a lubricated 6.0-mm cuffed tracheal tube over the bougie; rotate as it is advanced (prevents tip catching on arytenoids)
- Remove the bougie
- Inflate cuff and confirm ventilation with waveform capnography
- Secure the tube
Why Not Needle Cricothyroidotomy as First-Line?
- High failure rate in real-world emergencies (kinked catheter, misplacement, inadequate ventilation)
- Risk of barotrauma with jet insufflation
- Scalpel–bougie technique has superior success rates in simulation and case series
- Alternative scalpel–bougie techniques are also acceptable if performed competently
After eFONA
- Confirm ventilation with capnography
- Proceed to definitive airway management (formal tracheostomy if needed)
- Surgical cricothyroidotomy via eFONA should be converted to a formal tracheostomy within 24–72 hours once the patient is stable
SECTION 10: RAPID SEQUENCE INDUCTION AND INTUBATION (RSI)
Indication
- Full stomach (recent meal, bowel obstruction, trauma)
- Gastro-oesophageal reflux disease (symptomatic)
- Delayed gastric emptying (diabetics, opioid use, pain)
- Increased intra-abdominal pressure (pregnancy, obesity, ascites)
- Upper GI bleeding
Specific Considerations in RSI (2025 Updates)
- RSI patients frequently also have a physiologically difficult airway (haemodynamically unstable, hypoxic)
- These factors must be assessed and optimised before induction
- Remains an option in RSI but the 2025 guidelines acknowledge controversy
- Cricoid force may worsen laryngoscopic view — it should be removed if it obstructs intubation
- There is no robust evidence that cricoid pressure prevents aspiration; benefit must be balanced against harm
- Suxamethonium 1.5 mg/kg — standard for RSI (fastest onset and offset)
- Rocuronium 1.2 mg/kg — equivalent conditions if sugammadex available for reversal (CICO scenario)
- Do not attempt laryngoscopy before full NMB onset
- Traditional RSI avoids facemask ventilation to prevent gastric distension and aspiration
- However, in the hypoxic patient, gentle low-pressure facemask ventilation (PEEP < 10 cmH₂O) may be safer than proceeding to intubation with suboptimal oxygenation
- The 2025 guidelines support this modified RSI approach in selected patients
- HFNO during RSI (via nasal cannula under the facemask during preoxygenation) significantly extends safe apnoea time
- Same algorithm; same attempt limits; same escalation pathway
- eFONA may be anatomically more difficult in some RSI patients (obesity, distended abdomen causing neck extension difficulty)
SECTION 11: PHYSIOLOGICALLY DIFFICULT AIRWAY (New in 2025)
Definition and Rationale
- Hypoxaemia (SpO₂ < 93% on room air, or requiring supplemental O₂)
- Haemodynamic instability (hypotension, shock, vasopressor-dependent)
- Metabolic acidosis (pH < 7.2, severe sepsis, DKA, lactic acidosis)
- Severe respiratory failure (near-maximal respiratory effort, high FiO₂ requirements)
- Hypercapnia (pCO₂ > 6 kPa in a patient unable to tolerate even brief apnoea)
Why It Matters
- These patients desaturate faster and more severely than healthy patients
- Induction drugs may precipitate haemodynamic collapse
- Neuromuscular blockade removes all respiratory effort — if ventilation cannot be established, rapid deterioration follows
- The anaesthetic team may need to intubate in the ICU or emergency department, where conditions are less controlled
Management Principles
- Correct hypotension with fluids and vasopressors before induction where possible
- Optimise oxygenation — NIV or HFNO before intubation
- Correct reversible metabolic derangements where time allows
- Ensure full resuscitation team available
- HFNO is strongly recommended for peroxygenation
- Reduce time from induction to intubation
- Maximise first-pass success (VL, HFNO, adequate NMB)
- Plan for haemodynamic deterioration post-induction (vasopressor infusions prepared)
- Plan for haemodynamic support
- Sedation and ventilation strategy
- Avoid further oxygen desaturation
- Call for help earlier in physiologically difficult airway patients — expert assistance should be sought before the situation deteriorates
SECTION 12: OBESITY
Increased Risk
- Increased risk of difficult facemask ventilation (excess pharyngeal soft tissue)
- Increased risk of difficult SAD insertion and ventilation
- Potentially difficult tracheal intubation (restricted neck extension, soft tissue excess)
- More difficult eFONA (increased anterior neck soft tissue, CTM may be impalpable)
- Rapid haemoglobin oxygen desaturation due to:
- Reduced FRC
- Increased basal metabolic rate and O₂ consumption
- V/Q mismatch
- NAP4 and NAP7 both highlighted obesity as a major contributing factor to airway catastrophes
Recommendations (2025)
- Consider awake tracheal intubation (ATI) in patients with obesity when the airway is anticipated to be difficult
- Pre-oxygenate in head-up position (≥ 30°) — reduces atelectasis, improves FRC
- Consider performing airway management in the operating theatre (better equipment, positioning, lighting)
- HFNO for peroxygenation — strongly recommended; extends safe apnoea time
- Consider early use of a second-generation SAD if facemask ventilation is suboptimal (before proceeding through multiple Plan A attempts)
- Call for help earlier than in non-obese patients
- DAS 2025 explicitly aligns with the Society for Obesity and Bariatric Anaesthesia (SOBA) 2025 best-practice recommendations
SECTION 13: HUMAN FACTORS
Central Importance (Elevated in 2025)
Priming (Introduced 2025)
- After declared failure at Plan A → confirm eFONA kit accessible; identify who will perform it
- After maximum 3 SAD attempts → open the eFONA kit (assistant responsibility)
- Priming prevents the dangerous delay that occurs when teams scramble to find and set up eFONA equipment at the moment of crisis
Key Human Factors Concepts in 2025 Guidelines
| Concept | Practical Application |
|---|---|
| Shared mental model | All team members understand the plan before starting |
| Situational awareness | Continuously monitoring SpO₂, EtCO₂, patient colour |
| Cognitive offloading | Checklists reduce reliance on memory under stress |
| Clear role assignment | Designate intubator, bag-mask holder, eFONA performer in advance |
| Declaring failure | Explicit verbal declaration prevents anchoring and fixation |
| Stop-and-think prompts | Built into Plans B and C to prevent rushed decisions |
| Structured communication | PACE (Probe, Alert, Challenge, Emergency) for junior-to-senior escalation |
| Leadership and followership | Clear command structure; psychological safety to speak up |
| Debriefing | Mandatory after failed intubation and eFONA events |
SECTION 14: POINT-OF-CARE ULTRASOUND (POCUS)
New Endorsement in 2025
- Identify CTM and anterior neck anatomy — crucial for eFONA planning, especially in obese patients where CTM is impalpable
- Assess volume of anterior neck soft tissue (predictor of difficult intubation and difficult eFONA)
- Detect tracheal deviation or distortion
- Assess for airway masses
- Gastric ultrasound (antrum in right lateral decubitus position) allows estimation of gastric volume
- Can identify full stomach (liquid or solid content) before induction
- Informs RSI decision-making
- Tracheal intubation confirmation via trans-tracheal ultrasound — shows the tracheal tube sliding into the trachea in real time
- Complementary to waveform capnography (though capnography remains the gold standard)
SECTION 15: POST-INTUBATION CARE
Immediate Post-Intubation
- Confirm tracheal placement with waveform capnography (mandatory)
- Assess for complications: airway trauma, lip/dental injury, oropharyngeal lacerations
- Ensure haemodynamic stability (post-intubation hypotension is common, especially in physiologically difficult airways)
- Initiate appropriate ventilation strategy
After a Difficult Intubation or Failed Intubation Event
- Formulate an immediate post-procedure airway management plan — what will happen at extubation? Is re-intubation feasible if needed?
- Monitor for complications: subglottic oedema, haematoma, aspiration, laryngospasm, bronchospasm
- Complete airway alert form and any institutional incident reporting
Patient Communication
- Explain to the patient in person (post-procedure, when alert) and in writing that a difficult airway was encountered
- Provide the patient with a written airway alert card or letter describing the difficulty and equipment/technique used
- Send a written report to the patient's primary care provider with appropriate diagnostic coding (e.g., SNOMED CT 718446005)
Institutional Reporting
- Details should be entered into a national registry — DAS Difficult Airway Registry or DAS eFONA Registry
- Recorded using theatre coding systems for audit purposes
SECTION 16: DOCUMENTATION
Rationale
- Provide a reference for current and future airway management
- Inform case reviews and morbidity and mortality discussions
- Enable communication to other healthcare providers
What to Document
- Airway assessment findings
- Techniques and equipment used
- Difficulties encountered and what was attempted
- Outcomes
- Whether a technique was used for training or because of anticipated/actual difficulty (this distinction is important for future anaesthetists)
Communication After Difficult Airway
- Verbal handover to PACU/ICU/ward nursing staff regarding airway concerns
- Written documentation in the patient's record
- Verbal and written communication to the patient
- Written report to primary care provider
Recommendation
- Individuals have a professional responsibility to document airway management as required by their institution
SECTION 17: EDUCATION, TRAINING, AND INSTITUTIONAL RESPONSIBILITY
Philosophy
- Decision-making
- Communication
- Human factors
- Guideline implementation
- Multidisciplinary team training (not just the anaesthetist, but assistants, nurses, ODPs, surgeons)
Individual Responsibilities
- All anaesthetists should have regular training in scalpel cricothyroidotomy — this is non-negotiable
- Video laryngoscopy skills must be regularly practised
- Train in all steps of the algorithm A–D
- All airway managers should have training in capnography interpretation
Institutional Responsibilities
- Institutions must support clinicians to train with new equipment or guidelines
- Airway leads should reinforce familiarity with Plans A–D among all airway managers and assistants
- Human factors and team performance under stress must be included in training curricula
- Simulation is the cornerstone — regular, scenario-based training incorporating the full algorithm
Review and Governance
- Every failed intubation and every eFONA event should be reviewed and discussed at morbidity and mortality meetings
- Near-misses and complications must be reported and analysed
- Evidence of airway training can support annual performance assessments and revalidation/appraisal
COMPLETE ALGORITHM: DAS 2025 A–B–C–D at a Glance

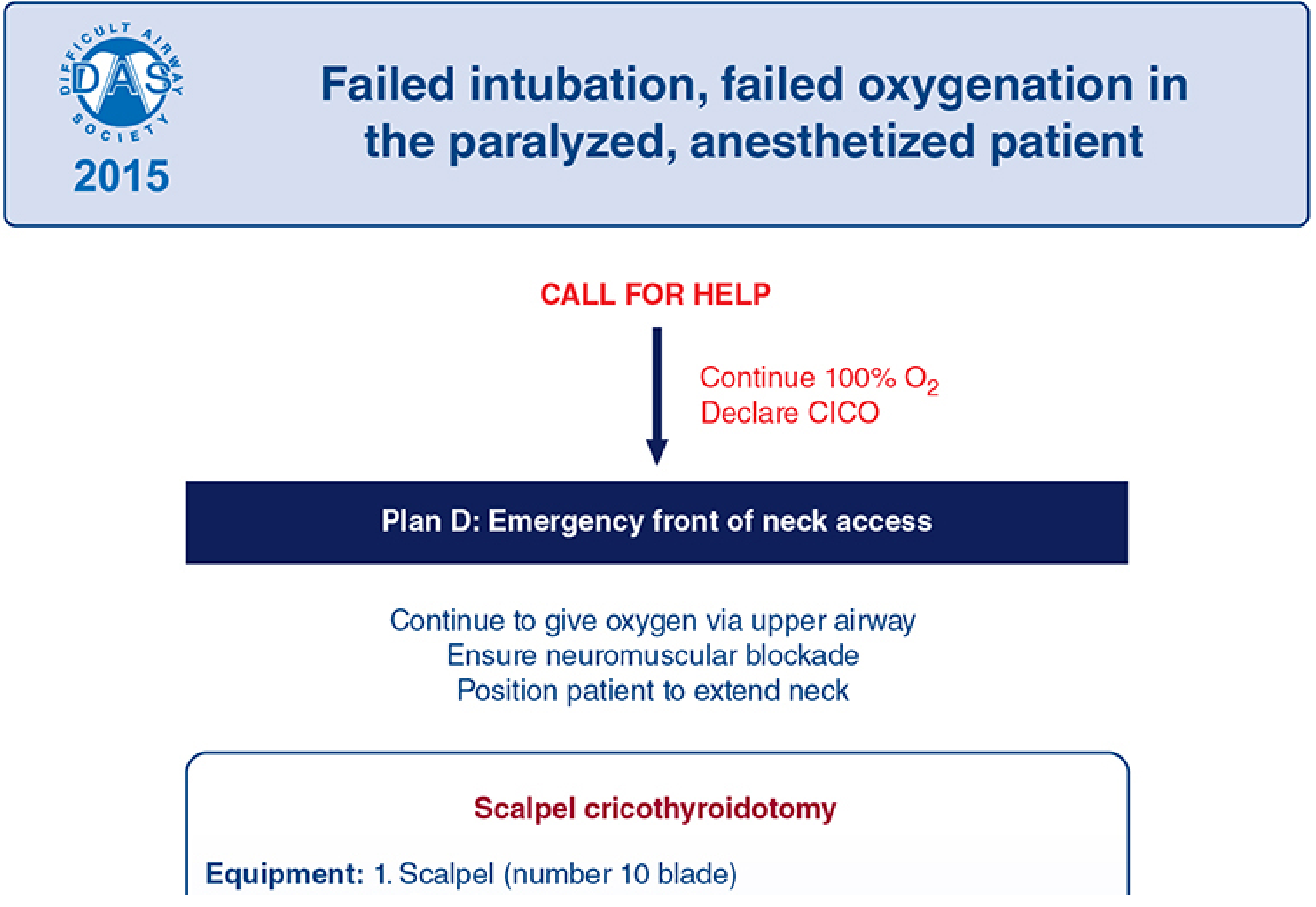
KEY DIFFERENCES: DAS 2015 vs DAS 2025
| Domain | DAS 2015 | DAS 2025 |
|---|---|---|
| Core philosophy | Manage failure | Optimise success |
| Plan A device | Direct or video laryngoscopy | Video laryngoscopy as default |
| Oxygenation | Recommended | Continuous, uninterrupted — central principle |
| HFNO | Not specified | Formally endorsed for peroxygenation |
| NMBAs | Conditional | Routinely recommended |
| Physiologically difficult airway | Not addressed | New dedicated section |
| Obesity | Brief mention | Dedicated section with specific recommendations |
| POCUS | Not mentioned | Formally endorsed — airway + gastric |
| Priming for eFONA | Reactive | Proactive, parallel to Plans A–C |
| "Wake up" option | Strongly preferred default | Context-dependent; not always possible |
| Human factors | Introduced as a section | Integral throughout the entire document |
| Quantitative NMB monitoring | Not specified | Recommended |
| Attempt limit (Plan A) | 3 + 1 | 3 + 1 maintained |
| Attempt limit (Plan B SAD) | 2 attempts with preferred device + 1 alternative | Maximum 3 attempts |
| Term "peroxygenation" | Not used | Newly coined, unified term |
| Documentation/registry | Recommended | DAS Difficult Airway Registry formally cited |
| Checklists | Not specified | Endorsed for emergency intubation |
SUMMARY OF ALL 65 RECOMMENDATIONS BY DOMAIN
| Domain | Number of Recommendations | Key Themes |
|---|---|---|
| Airway assessment | 3 | History, exam, bedside tests, physiological assessment |
| Planning and strategy | 9 | Plans A–D addressed, awake intubation, communication, location, checklists |
| Monitoring | 5 | Capnography, SpO₂ tones, quantitative NMB monitoring |
| Drugs | 1 | NMBAs routinely for intubation |
| Peroxygenation | 3 | Head-up, positive pressure, HFNO for at-risk patients |
| Plan A | ~8 | VL first-line, 3+1, meaningful changes, capnography, declare failure |
| Plan B | ~7 | Second-generation SAD, 3 attempts, Stop and Think, priming |
| Plan C | ~4 | Two-person, adjuncts, NMB, declare CICO |
| Plan D (eFONA) | ~6 | Scalpel–bougie–tube, technique steps, priming, all should train |
| RSI | ~4 | Aspiration risk, modified RSI, physiological assessment |
| Physiologically difficult airway | ~4 | Pre-optimisation, HFNO, call early for help |
| Obesity | 5 | ATI consideration, head-up, HFNO, early SAD, SOBA alignment |
| Human factors | ~5 | Priming, shared mental model, checklists, PACE, debriefing |
| POCUS | ~3 | Airway assessment, gastric assessment, confirmation |
| Documentation | 1 | Professional responsibility; airway alert; national registry |
| Education and training | ~7 | eFONA training, VL, capnography, simulation, M&M review, appraisal |