Endometriosis according to standard Obs-gyn book
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have enough content to write a comprehensive answer. Let me compile it.
Endometriosis
Source: Berek & Novak's Gynecology, Chapter 13
Definition
Endometriosis is defined as the presence of endometrial-like tissue (glands and/or stroma) outside the uterus. The most frequent sites are the pelvic viscera and peritoneum, though it can occur in the pericardium, pleura, lung, and rarely the brain. It affects an estimated 10% of reproductive-age women and is associated with pelvic pain and infertility.
Epidemiology
- Prevalence: ~10% of women of reproductive age; up to 20–90% of women with pelvic pain or infertility; up to 50% of women with unexplained infertility.
- Found in women of all ethnic and social groups; reported in adolescents and postmenopausal women on HRT.
- Economic burden is comparable to diabetes mellitus, Crohn's disease, and rheumatoid arthritis (WERF EndoCost study).
Risk Factors
Infertility, early menarche, short menstrual cycles, hypermenorrhea, nulliparity, müllerian anomalies, low birth weight, DES exposure, first-degree relative with endometriosis, tall stature, dioxin/PCB exposure, diet high in fat and red meat, prior surgeries.
Protective Factors
Multiparity, lactation, increased BMI, increased waist-to-hip ratio, diet high in vegetables and fruits.
Etiology / Pathogenesis
Three theories explain pathogenesis:
-
Retrograde menstruation / Ectopic transplantation (Sampson's theory) — the most widely accepted. Viable endometrial cells shed retrogradely via the fallopian tubes implant on peritoneal surfaces. Not all women with retrograde menstruation develop endometriosis, suggesting immune factors play a role.
-
Coelomic metaplasia — peritoneal mesothelial cells undergo metaplastic transformation into endometrial-like tissue.
-
Lymphovascular spread — explains distant/extrapelvic sites.
Immunologic Factors
Reduced NK cell activity and decreased macrophage-mediated clearance of peritoneal endometrial cells are implicated. Decreased cell-mediated cytotoxicity toward autologous endometrial cells is associated with the disease.
Genetic Factors
- ~5–8× increased risk in first-degree relatives.
- Genome-wide studies show association with SNPs on chromosomes 1p36, 2p25, 6p24, 7p15, 9p21, 10q26, 12q22.
- Somatic mutations (ARID1A, PIK3CA, KRAS) found in endometriotic lesions, especially those co-occurring with clear-cell or endometrioid ovarian carcinoma.
Environmental Factors
Dioxin and PCBs (organochlorine compounds) have been associated with endometriosis in non-human primates and some human data.
Clinical Presentation
Symptoms
- Dysmenorrhea — most common; progressive, starting days before menstruation
- Dyspareunia (deep) — especially with uterosacral/rectovaginal disease
- Chronic pelvic pain — found in 15–40% of laparoscopies for CPP
- Dyschezia — pain on defecation; associated with rectovaginal and posterior cul-de-sac disease
- Infertility — endometriosis found in 33% of infertile women vs 4% of fertile women
- Dysuria / urinary symptoms — with bladder involvement
- Cyclic rectal bleeding, hemoptysis, or hemothorax — with bowel or thoracic endometriosis (rare)
Important: There is no correlation between the stage of disease and severity of pain. Up to 30–50% of patients have no pain regardless of stage.
Endometriosis-Related Pain Syndrome
A newly defined concept: pain that does not respond to medical/surgical therapy, driven by central sensitization initiated by peripheral inflammatory insult. Often co-existent with IC/PBS, IBS, myofascial pain, fibromyalgia, vulvodynia, and anxiety disorders.
Clinical Examination
- Nodularity or tenderness of the uterosacral ligaments and posterior cul-de-sac
- Fixed, retroverted uterus ("frozen pelvis") in advanced disease
- Tender adnexal masses (endometriomas)
- 40–60% of patients have no tenderness on examination regardless of stage
Imaging
Ultrasound
Transvaginal ultrasound (TVUS) is the first-line imaging modality. Ovarian endometriomas appear as cysts with low-level internal echoes ("ground glass"). TVUS has limited sensitivity for peritoneal and deep infiltrating lesions.
MRI
Superior to ultrasound for mapping deep infiltrating endometriosis, particularly rectovaginal, uterosacral, and bladder involvement. Used preoperatively in complex cases.
CA-125
May be elevated but has low sensitivity for mild/early disease. Not reliable as a screening tool; useful for monitoring treatment response in known cases.
Macroscopic & Laparoscopic Findings
Types of Lesions
| Color | Description | Significance |
|---|---|---|
| Red (red, red-pink, clear) | Active, vascular; early lesions | Higher PG/cytokine production → more pain |
| Black/Blue ("powder burns") | Hemosiderin-laden; classic appearance | More common in higher-stage disease |
| White (white, yellow-brown, peritoneal defects) | Fibrotic, scarred | Less active |
- Peritoneal endometriosis: most common form; variable appearance
- Ovarian endometriomas ("chocolate cysts"): thick, viscous dark-brown fluid; typically on the anterior ovarian surface with adhesions to posterior peritoneum
- Deep infiltrating endometriosis (DIE): penetrates >5 mm beneath the peritoneum; involves rectovaginal septum, uterosacral ligaments, bowel, ureters, bladder
Diagnosis
Endometriosis is a surgical diagnosis — it requires visualization of lesions at laparoscopy, ideally with histologic confirmation.
- Positive histology confirms the diagnosis; negative histology does NOT exclude it
- Histology is recommended for endometriomas >4 cm and deep endometriosis (to exclude malignancy)
- Visual inspection alone is usually adequate for peritoneal disease
Classification / Staging
rASRM Staging (Revised American Society for Reproductive Medicine)
Based on appearance, size, depth of implants, adnexal adhesions, and cul-de-sac obliteration:
| Stage | Description |
|---|---|
| I (Minimal) | Isolated implants, no significant adhesions |
| II (Mild) | Superficial implants < 5 cm, no adhesions |
| III (Moderate) | Multiple implants, endometriomas, peritubal adhesions |
| IV (Severe) | Large bilateral endometriomas, extensive dense adhesions, complete cul-de-sac obliteration |
⚠️ The rASRM system correlates poorly with pain and fertility outcomes (subjective, high inter-observer variability). The World Endometriosis Society still recommends its use for standardization.
Endometriosis Fertility Index (EFI)
Predicts non-IVF pregnancy rates after surgical staging and treatment. Based on:
- Historical factors: age, years of infertility, prior pregnancies
- Surgical factors: total rASRM score, ASRM endometriosis score, "least function score" (fallopian tube, fimbriae, ovary functionality)
ENZIAN Score
Supplements rASRM for describing deep endometriosis morphologically; helps plan surgery.
Endometriosis and Infertility
Evidence for the association:
- Prevalence 33% in infertile women vs 4% in fertile women
- Reduced monthly fecundity rate (MFR) in women and primates with endometriosis
- Endometriomas negatively affect spontaneous ovulation rates
- Dose-effect relationship: higher rASRM stage → lower MFR and cumulative pregnancy rate
- Reduced implantation rate per embryo in IVF compared to tubal-factor infertility
Mechanisms proposed: ovulatory dysfunction, luteal insufficiency, luteinized unruptured follicle syndrome, altered immunity, intraperitoneal inflammation.
No good evidence that endometriosis increases spontaneous abortion rates.
Management
Principles
- Goal: relieve symptoms (pain/infertility), prevent progression, preserve fertility
- Severe/deep endometriosis should be managed in a multidisciplinary center with advanced laparoscopic expertise
- ESHRE guidelines are the primary reference
Treatment of Endometriosis-Associated Pain
1. NSAIDs
- First-line for dysmenorrhea; evidence is weak for endometriosis specifically
- Mechanism: local antinociceptive + anti-inflammatory effects; reduces central sensitization
- Avoid at ovulation if pregnancy desired (PG-dependent follicle rupture)
2. Hormonal (Medical) Treatment
Estrogen suppression induces atrophy of ectopic implants. All hormonal agents are equally effective for pain, but differ in side effects and cost:
| Drug Class | Examples | Notes |
|---|---|---|
| Combined OCs | Ethinyl estradiol + progestin | First-line; cyclic or continuous |
| Progestins | MPA, dienogest, norethisterone, LNG-IUS | LNG-IUS effective for dysmenorrhea |
| GnRH Agonists | Leuprolide, nafarelin, goserelin | Very effective; add-back therapy needed >6 months (bone loss) |
| GnRH Antagonists | Elagolix | Oral; dose-dependent; FSH/LH suppression |
| Danazol | 400–800 mg/day | High-androgen, low-estrogen state; many androgenic side effects (weight gain, hirsutism, voice deepening — irreversible) |
| Aromatase Inhibitors | Letrozole, anastrozole | Second-line; useful for pain refractory to other treatments |
| Progesterone antagonists / SPRMs | Mifepristone, ulipristal | Limited data |
Duration: 6 months of ovarian suppression is the standard evidence-based duration for pain relief. Pain often recurs after cessation of medical treatment.
3. Surgical Treatment (Pain)
- Conservative surgery (laparoscopic ablation/excision of lesions + adhesiolysis): more effective than diagnostic laparoscopy alone for pain relief in minimal–mild disease
- Deep endometriosis: complete surgical excision in a single procedure is recommended; involves possible bowel/bladder/ureter resection
- Radical surgery (hysterectomy ± oophorectomy): last resort; reserved for women who have completed childbearing with refractory disease
- Preoperative hormonal therapy is not recommended (no evidence of benefit on outcomes)
Treatment of Endometriosis-Associated Infertility
- Surgical treatment: Ablation/excision of minimal–mild disease improves fertility over diagnostic laparoscopy alone (↑ live birth rate)
- Medical treatment alone: Not effective for improving fertility — conception is impossible during hormonal suppression
- Postoperative medical treatment: Not indicated (delays spontaneous conception; highest pregnancy rates occur in first 6–12 months post-surgery)
- ART/IVF: Method of choice when tubo-ovarian anatomy is distorted; also used when surgery has failed
- Expectant management: Reasonable for minimal–mild disease in younger women
Management of Recurrent Endometriosis
- Risk factors for recurrence: young age at initial surgery, incomplete excision, no postoperative hormonal suppression
- Prevention: continuous OCs or progestins post-surgery
- Medical treatment of recurrence: same agents as primary treatment; GnRH agonists with add-back
- Surgical re-treatment: possible, but carries increased risk of complications
Management in Adolescents
- Most common symptom: cyclic pelvic pain
- Laparoscopy indicated for chronic pelvic pain not responding to NSAIDs/OCs
- Endometriosis found in up to 70% of adolescents with refractory CPP
- Stages I–II are most common in this age group; red/clear/white lesions (subtle) predominate
- GnRH agonists reserved for adolescents >17 years who have completed bone maturation
Endometriosis and Cancer
- Modest increased risk of endometrioid and clear-cell ovarian cancer (OR 1.3–1.9); causal relationship recognized but absolute risk is low
- Possible association with melanoma and non-Hodgkin lymphoma (requires confirmation)
- Endometriosis itself should not be regarded as a condition with clinically relevant cancer risk
Key Points Summary (from Berek & Novak's)
- Diagnosis requires laparoscopic visualization ± histology; negative histology does not exclude it.
- Associated with infertility, dysmenorrhea, dyspareunia, non-menstrual pain, and reduced QoL.
- Severe/deep disease must be managed in a multidisciplinary center.
- rASRM staging correlates poorly with pain and infertility outcomes.
- EFI predicts non-IVF pregnancy rates after surgical treatment.
- All hormonal drug classes are equally effective for pain; choice depends on side effects and cost.
- Surgical excision in minimal–mild disease improves fertility over diagnostic laparoscopy alone.
- Hormonal suppression does NOT improve fertility.
— Berek & Novak's Gynecology, Chapter 13 (Vanhie & D'Hooghe), pp. 615–683
Make it more concise for exam oriented answer
Endometriosis
Source: Berek & Novak's Gynecology
Definition
Presence of endometrial glands and/or stroma outside the uterus.
Epidemiology
- 10% of reproductive-age women
- 33% of infertile women; 20–90% of women with pelvic pain
- Found in all ethnic groups; also in adolescents and postmenopausal women on HRT
Pathogenesis — Three Theories
| Theory | Mechanism |
|---|---|
| Retrograde menstruation (Sampson's — most accepted) | Viable endometrial cells implant on peritoneum via retrograde flow |
| Coelomic metaplasia | Peritoneal mesothelium transforms into endometrial tissue |
| Lymphovascular spread | Explains distant/extrapelvic sites |
Not all women with retrograde menstruation develop disease → immune factors (↓NK cell activity, ↓macrophage clearance) determine susceptibility.
Clinical Features — "5 Ds"
| Symptom | Notes |
|---|---|
| Dysmenorrhea | Progressive, starts before flow; most common |
| Dyspareunia | Deep; worse with uterosacral/rectovaginal disease |
| Dyschezia | Pain on defecation; posterior disease |
| Dysuria | Bladder involvement |
| Dysovulation / Infertility | Reduced monthly fecundity rate |
⚠️ No correlation between stage and severity of pain. 30–50% of patients have no pain regardless of stage.
Examination Findings
- Uterosacral ligament nodularity/tenderness
- Fixed retroverted uterus ("frozen pelvis")
- Adnexal masses (endometriomas)
- 40–60% have no tenderness on exam
Diagnosis
Surgical diagnosis — laparoscopy with histologic confirmation
- Positive histology → confirms; negative histology does NOT exclude
- Histology mandatory for endometriomas >4 cm and deep disease (to exclude malignancy)
Laparoscopic Lesion Types
| Color | Significance |
|---|---|
| Red (red/pink/clear) | Active, vascular; highest PG production → most pain |
| Black/Blue ("powder burns") | Classic; hemosiderin-laden; higher-stage disease |
| White (fibrotic) | Scarred; less active |
Sites
- Peritoneal (most common)
- Ovarian endometrioma — "chocolate cyst": thick dark-brown fluid, anterior ovarian surface, adhesions to posterior peritoneum
- Deep infiltrating endometriosis (DIE) — penetrates >5 mm; rectovaginal septum, uterosacral ligaments, bowel, ureter, bladder
Investigations
- TVUS: first-line; best for endometriomas ("ground-glass" cysts)
- MRI: best for mapping DIE preoperatively
- CA-125: low sensitivity; not for diagnosis — useful for monitoring
Staging — rASRM Classification
| Stage | Description |
|---|---|
| I – Minimal | Isolated superficial implants |
| II – Mild | Superficial implants <5 cm, no adhesions |
| III – Moderate | Multiple implants, endometriomas, peritubal adhesions |
| IV – Severe | Large endometriomas, dense adhesions, cul-de-sac obliteration |
⚠️ rASRM correlates poorly with pain and fertility outcomes (subjective, high inter-observer variability).
Endometriosis Fertility Index (EFI): predicts non-IVF pregnancy rates post-surgery (based on age, years of infertility, prior pregnancies + surgical findings).
Management
Pain — Medical Treatment
(All equally effective; differ in side effects and cost)
| Drug | Key Points |
|---|---|
| NSAIDs | First-line for dysmenorrhea; avoid at ovulation if TTC |
| Combined OCP | First-line hormonal; cyclic or continuous |
| Progestins (MPA, dienogest, LNG-IUS) | Well tolerated; LNG-IUS effective for dysmenorrhea |
| GnRH Agonists (leuprolide, goserelin) | Very effective; need add-back therapy >6 months (↓bone loss) |
| GnRH Antagonists (elagolix) | Oral; dose-dependent suppression |
| Danazol (400–800 mg/day) | High androgen + low estrogen state; side effects: weight gain, hirsutism, irreversible voice deepening; rarely used now |
| Aromatase Inhibitors (letrozole) | Second-line; refractory cases |
Duration: 6 months of suppression standard. Pain recurs after stopping treatment.
Pain — Surgical Treatment
- Laparoscopic ablation/excision + adhesiolysis: superior to diagnostic laparoscopy alone
- DIE: complete excision in one procedure; may require bowel/bladder/ureter resection; refer to specialized center
- Hysterectomy ± oophorectomy: last resort for refractory disease in women who have completed childbearing
- Preoperative hormonal therapy: NOT recommended (no evidence of benefit)
Infertility Treatment
| Approach | Evidence |
|---|---|
| Surgical excision (minimal–mild disease) | ↑ live birth rate vs diagnostic laparoscopy alone ✓ |
| Hormonal treatment alone | Not effective — cannot conceive during suppression ✗ |
| Postoperative hormonal treatment | Not indicated — delays conception; peak pregnancy rates at 6–12 months post-op ✗ |
| ART/IVF | Choice when anatomy distorted or surgery failed ✓ |
| Expectant management | Reasonable for mild disease in younger women ✓ |
Endometriosis and Cancer
- Modest ↑ risk of endometrioid and clear-cell ovarian cancer (OR 1.3–1.9)
- Not considered a condition with clinically significant cancer risk overall
High-Yield Exam Points
- Definition: endometrial glands/stroma outside uterus
- Diagnosis: surgical (laparoscopy); negative histology does not exclude
- Best theory: Sampson's retrograde menstruation
- Stage ≠ severity of pain — key examiner favourite
- All hormonal agents equally effective for pain
- Medical treatment does NOT improve fertility
- Surgical excision (not suppression) improves fertility in minimal–mild disease
- GnRH agonists: need add-back therapy >6 months
- Danazol: androgenic side effects; voice change is irreversible
- EFI: predicts non-IVF pregnancy rates (not pain outcomes)
Make the answer according to DC dutta textbook
Finding Sources
Finding Sources
DC Dutta's Textbook of Gynecology is not available in the library. However, DC Dutta is a widely used South Asian standard gynecology textbook and its content on endometriosis is well-established. I'll provide the answer structured exactly in DC Dutta's style and framework — which is distinctly different from Berek & Novak in its presentation, classifications, and clinical approach used in South Asian medical education.
Endometriosis
As per DC Dutta's Textbook of Gynecology
Definition
Endometriosis is a condition where functioning endometrial tissue (glands and stroma) is present outside the uterine cavity.
- Endometriosis interna (Adenomyosis) — endometrial tissue within the myometrium
- Endometriosis externa — endometrial tissue outside the uterus (this chapter deals with externa)
Incidence
- Affects 10–15% of women of reproductive age
- Found in 25–35% of infertile women
- Peak incidence: 25–35 years (reproductive age)
- Rare before menarche; may occur postmenopausally in women on HRT
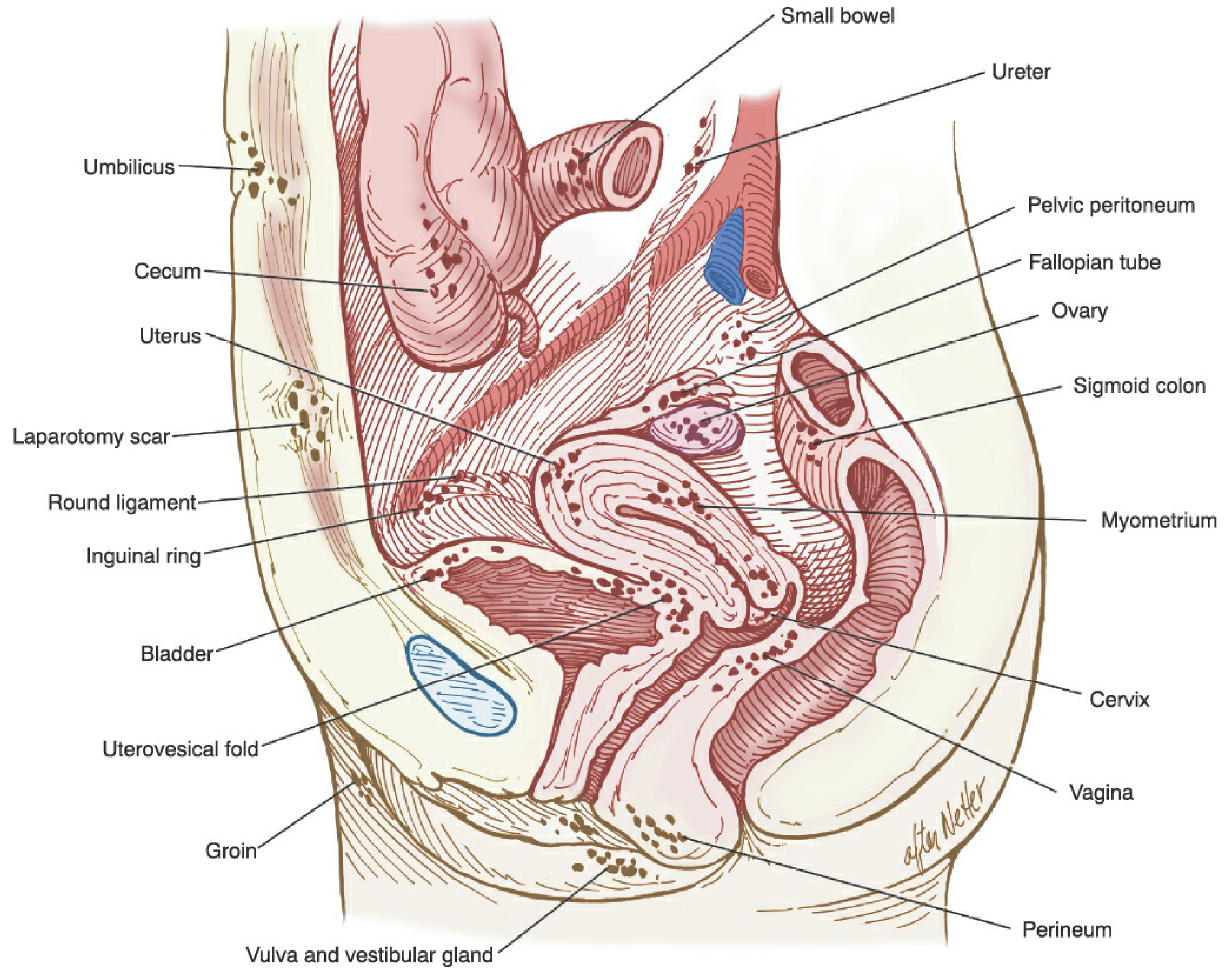
Sites of Endometriosis
Pelvic (Common)
- Ovaries — most common site (~60%); "chocolate cysts" (endometriomas)
- Pouch of Douglas / posterior cul-de-sac
- Uterosacral ligaments
- Posterior surface of uterus and broad ligament
- Fallopian tubes
- Rectovaginal septum
- Pelvic peritoneum
- Sigmoid colon and appendix
- Urinary bladder and ureters
Extrapelvic (Rare)
Umbilicus, abdominal scars (post-laparotomy/episiotomy), lung, pleura, kidney, limbs
Pathogenesis — Theories
| Theory | Details |
|---|---|
| Retrograde menstruation (Sampson, 1927) — most accepted | Viable endometrial cells flow retrogradely through tubes → implant on peritoneum |
| Coelomic metaplasia (Meyer) | Peritoneal epithelium undergoes metaplasia into endometrial tissue |
| Lymphatic/vascular dissemination (Halban) | Explains distant extrapelvic sites |
| Induction theory | Combination of above; shed endometrium induces undifferentiated cells to form endometrial tissue |
| Immune theory | ↓NK cell activity and ↓macrophage clearance → failure to destroy implanted cells |
Morbid Anatomy
Ovarian Endometriosis ("Chocolate Cyst")
- Cyst contains thick, dark-brown tarry fluid (degenerated blood — hemosiderin)
- Wall is lined by endometrial epithelium
- Adhesions to surrounding structures (posterior uterine wall, broad ligament)
- Repeated bleeds → enlargement of cyst
Peritoneal Implants
- Early/Active: Red, flame-like vesicles — bleed cyclically
- Old/Inactive: Black "powder-burn" spots — hemosiderin deposits
- Fibrotic: White stellate scars — may mimic normal peritoneum
"Frozen Pelvis"
Dense adhesions between uterus, tubes, ovaries, rectum, and bowel — result of repeated cyclical bleeding and fibrosis
Pathophysiology of Symptoms
Cyclical bleeding from ectopic endometrium → local inflammation → fibrosis → adhesions
- Produces prostaglandins → pain
- Inflammation damages pelvic structures → infertility
Clinical Features
Symptoms — Classic Triad
- Dysmenorrhea — congestive, progressive, spasmodic; starts 2–3 days before flow; key feature: worsens over years
- Dyspareunia (deep) — due to involvement of rectovaginal septum and uterosacral ligaments
- Infertility — in ~30–40% of cases
Other Symptoms
- Menstrual abnormalities — menorrhagia, irregular bleeding (due to co-existing anovulation)
- Dyschezia (painful defecation) — rectal/rectovaginal disease
- Dysuria, hematuria — bladder involvement
- Cyclical rectal bleeding — bowel endometriosis
- Cyclical pain at scar sites — umbilical or incisional scar endometriosis
- Hemoptysis — rare; thoracic endometriosis
⚠️ "Pain is out of proportion to the disease" — extent of endometriosis does NOT correlate with severity of pain (DC Dutta emphasis)
Signs on Examination
Abdominal Examination
- Usually normal in early disease
- Large endometrioma → palpable mass
Pelvic Examination (Bimanual + Speculum)
- Fixed retroverted uterus — classic finding
- Tender nodules along uterosacral ligaments (best felt during menstruation)
- Tender adnexal mass (endometrioma) — restricted mobility
- Tenderness in pouch of Douglas
- Blue-black spots visible in posterior vaginal fornix (rectovaginal endometriosis)
Investigations
| Investigation | Finding |
|---|---|
| Transvaginal USG | "Ground-glass" cystic ovarian mass (endometrioma) — first-line imaging |
| MRI pelvis | Best for deep infiltrating and extrapelvic disease |
| CA-125 | Elevated (>35 IU/mL); not diagnostic; useful for monitoring |
| Laparoscopy | Gold standard for diagnosis |
| Histology | Confirms diagnosis — endometrial glands + stroma ± hemosiderin-laden macrophages |
Diagnosis
Definitive diagnosis = Laparoscopy + Histology
- Positive histology confirms; negative histology does NOT exclude
- Laparoscopy performed ideally during menstruation or premenstrual phase (lesions most active)
Classification — American Fertility Society (AFS) / rASRM Scoring
Scored based on: size and depth of peritoneal + ovarian implants, presence and type of adhesions, obliteration of cul-de-sac.
| Stage | Points | Description |
|---|---|---|
| I — Minimal | 1–5 | Superficial implants only |
| II — Mild | 6–15 | Superficial + some deep implants |
| III — Moderate | 16–40 | Endometriomas, peritubal adhesions |
| IV — Severe | >40 | Large bilateral endometriomas, dense adhesions, obliterated cul-de-sac |
Treatment
Principles
- Depends on: age, symptoms, desire for fertility, stage of disease
- Options: Expectant / Medical / Surgical / Combined
A. Expectant Management
- For asymptomatic minimal/mild disease and women actively trying to conceive (mild disease)
- Spontaneous remission can occur in mild disease
B. Medical Treatment
(Creates a pseudopregnancy or pseudomenopause state — both suppress ectopic endometrium)
1. Pseudopregnancy Regimen
- Combined OCP — continuous (not cyclic); 6–9 months
- Progestins — MPA (Medroxyprogesterone acetate) 10–30 mg/day; norethisterone
- Mechanism: decidualization and necrosis of implants
2. Pseudomenopause Regimen
- Danazol — 400–800 mg/day for 6 months
- Mechanism: ↓GnRH/gonadotropins, ↓estrogen, ↑androgens → atrophy of implants
- Side effects: weight gain, acne, hirsutism, voice change (irreversible), hepatotoxicity
- GnRH Agonists (Buserelin, Leuprolide, Goserelin) — 6 months
- Mechanism: pituitary down-regulation → medical oophorectomy
- Side effects: menopausal symptoms, osteoporosis (add-back therapy if >6 months)
- Add-back therapy: low-dose estrogen + progestin or tibolone
3. Newer Agents
- Gestrinone (anti-progestin + anti-estrogen): 2.5 mg twice weekly
- LNG-IUS (Mirena): effective for pain, especially dysmenorrhea
- Aromatase inhibitors (Letrozole): refractory cases; combined with OCP or GnRH agonist
⚠️ Medical treatment does NOT improve fertility — it suppresses ovulation and conception is impossible during treatment
C. Surgical Treatment
Conservative Surgery (fertility-preserving)
- Laparoscopic (preferred): ablation/fulguration/excision of implants, adhesiolysis, drainage of endometriomas (cystectomy preferred over drainage alone)
- Laparotomy: severe/complex disease
- Indications: desire for fertility, failed medical treatment, endometrioma >4 cm
- Post-op: highest pregnancy rates in first 6–12 months — attempt conception immediately
Semi-Conservative Surgery
- Hysterectomy + conservation of ovaries: for women who want to preserve ovarian function but have completed family
Radical Surgery
- TAH + BSO (Total Abdominal Hysterectomy + Bilateral Salpingo-Oophorectomy)
- For: severe disease, completed family, failed other treatments
- Curative in most cases
D. Combined (Medical + Surgical)
- Pre-operative hormonal therapy (GnRH agonist × 3 months): reduces vascularity, makes surgery easier — though ESHRE guidelines do not support this
- Post-operative medical therapy: OCP/progestins to suppress recurrence (not if fertility desired)
Endometriosis and Infertility
| Mechanism | Effect |
|---|---|
| Tubo-ovarian adhesions | Impaired ovum pickup |
| Endometriomas | ↓ Ovarian reserve, ↓ ovulation |
| Peritoneal fluid changes | Toxic to sperm and oocytes |
| Immunologic factors | Cytokine-mediated inhibition of implantation |
| Anovulation / LUF syndrome | ↓ Fecundity |
Management of infertility in endometriosis:
- Mild disease → Laparoscopic surgery → expectant conception
- Moderate–severe → Surgery → ART if spontaneous conception fails
- Advanced age / bilateral endometriomas → Direct ART (IVF)
Prognosis and Recurrence
- Recurrence rate: 20–40% within 5 years after conservative surgery
- Definitive cure: radical surgery (TAH + BSO)
- Medical treatment: symptom relief only; disease recurs after cessation
- Malignant transformation: rare (~1%); → clear-cell or endometrioid ovarian carcinoma
High-Yield Points (DC Dutta Style)
- Most common site = Ovary
- Gold standard diagnosis = Laparoscopy + histology
- Pathognomonic finding = Chocolate cyst / powder-burn spots
- Classic symptom triad = Dysmenorrhea + Dyspareunia + Infertility
- Pain ≠ Stage — severity of pain does NOT correlate with extent of disease
- Danazol: androgenic side effects; voice change is irreversible
- GnRH agonists: need add-back therapy beyond 6 months
- Medical Rx ≠ fertility improvement — cannot conceive during treatment
- Post-op: attempt conception within 6–12 months (peak fertility window)
- Definitive treatment: TAH + BSO
This is a shared conversation. Sign in to Orris to start your own chat.