Seventh cranial nerve
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
facial nerve anatomy course branches CN7
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Now I have rich, comprehensive content. Let me compile the full answer.
Facial Nerve (CN VII) — The Seventh Cranial Nerve
Fiber Composition
The facial nerve is a mixed nerve with five functional fiber types:
| Fiber Type | Function |
|---|---|
| Special Visceral Efferent (SVE / Branchial efferent) | Motor to muscles of facial expression, stapedius, stylohyoid, posterior digastric |
| General Visceral Efferent (GVE / Parasympathetic) | Secretomotor to lacrimal gland, nasal/palatine glands (via GSPN), submandibular & sublingual glands (via chorda tympani) |
| Special Afferent (SA / Taste) | Taste — anterior 2/3 of tongue (chorda tympani), palate & tonsillar fossa (GSPN) |
| General Somatic Afferent (GSA) | Sensation from skin of external auditory canal and auricle |
| General Visceral Afferent (GVA) | Mucosa of nose, pharynx, palate |
Brainstem Nuclei
Three brainstem nuclei correspond to these modalities:
- Motor nucleus of CN VII (pons) → SVE fibers
- Superior salivatory nucleus (pons) → GVE (parasympathetic) fibers
- Nucleus of the solitary tract (pons/medulla) → receives taste (SA) and visceral afferent (GVA) input
The motor fibers have a distinctive intracranial loop — they curve around the abducens nucleus (CN VI), forming the facial colliculus visible on the floor of the fourth ventricle.
Course
1. Posterior Cranial Fossa
Emerges at the pontomedullary junction as two roots:
- A large motor root
- A smaller nervus intermedius (sensory + parasympathetic root)
Both cross the posterior cranial fossa and enter the internal acoustic meatus (IAM) of the petrous temporal bone, alongside CN VIII and the labyrinthine artery.
2. Within the Temporal Bone — The Fallopian (Facial) Canal
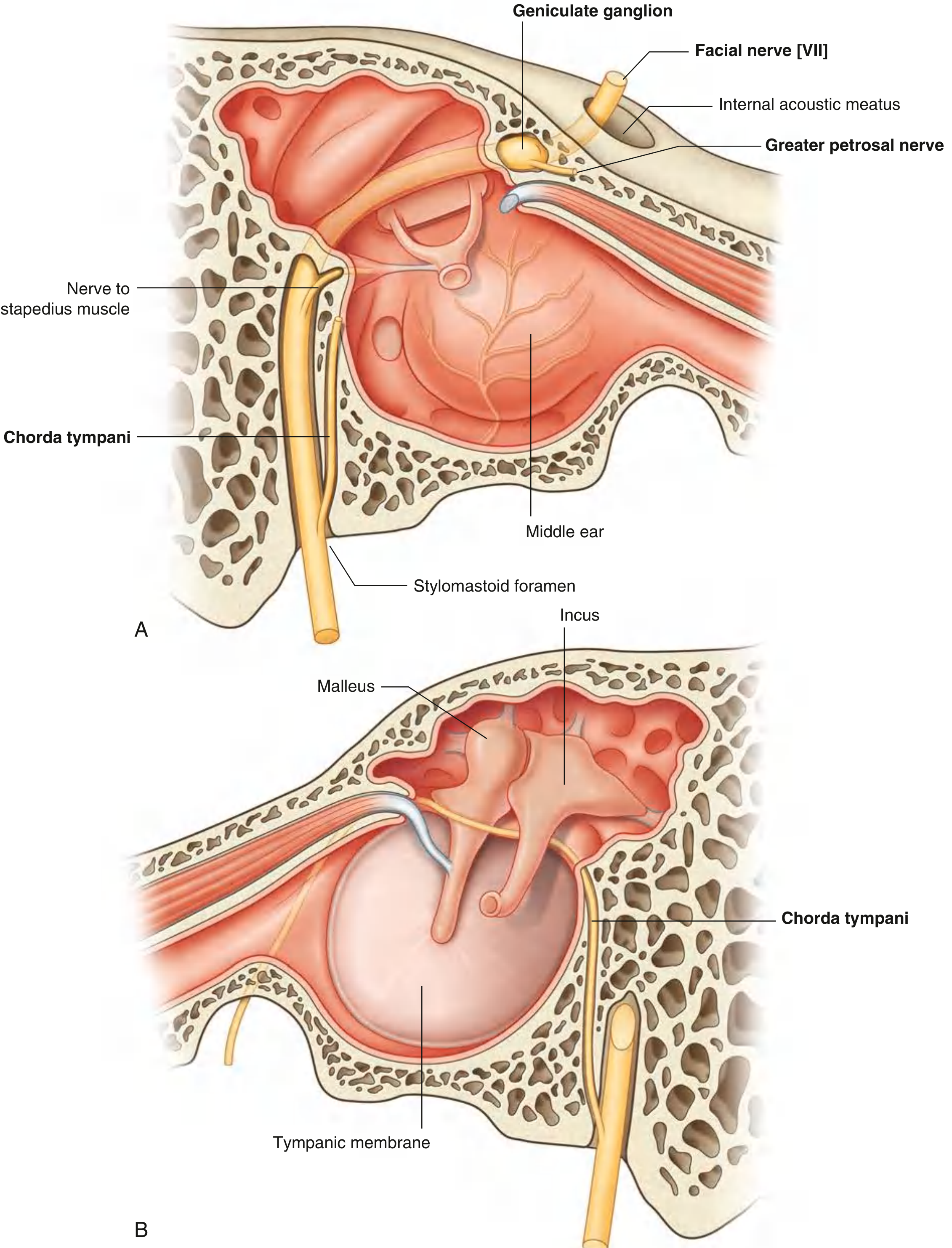
At the distal IAM, the two roots fuse and the nerve enters the fallopian canal through three segments:
| Segment | Location | Features |
|---|---|---|
| Labyrinthine | Fundus of IAC → geniculate ganglion | Shortest, narrowest; no fibrous sheath; runs superior to cochlea |
| Tympanic (horizontal) | Geniculate ganglion → pyramidal eminence | 1st genu (sharp posterior turn); bulges on medial wall of middle ear; overlies oval window |
| Mastoid (vertical) | Pyramidal eminence → stylomastoid foramen | 2nd genu at pyramidal eminence; gives off nerve to stapedius and chorda tympani |
The nerve exits the skull through the stylomastoid foramen.
3. Extracranial Course
After exiting the stylomastoid foramen, the nerve enters the parotid gland, divides into upper (temporofacial) and lower (cervicofacial) trunks, and ramifies into five terminal branches that emerge from the borders of the gland:
- Temporal — frontalis, orbicularis oculi (upper)
- Zygomatic — orbicularis oculi (lower), zygomaticus
- Buccal — buccinator, orbicularis oris
- Marginal mandibular — depressors of lip/chin
- Cervical — platysma
(Mnemonic: Two Zealous Bobbies Make Coffee — or "Ten Zebras Bit My Calf")
Branches and Their Functions
| Branch | Origin | Carries | Function |
|---|---|---|---|
| Greater petrosal nerve (GSPN) | Geniculate ganglion | Preganglionic parasympathetic + taste | → pterygopalatine ganglion → lacrimal gland, nasal/palatine glands |
| Nerve to stapedius | Mastoid segment | Motor | Dampens ossicular chain (lesion → hyperacusis) |
| Chorda tympani | Mastoid segment, just above stylomastoid foramen | Preganglionic parasympathetic + taste | Crosses middle ear between malleus and incus; exits via petrotympanic fissure; joins lingual nerve → submandibular ganglion, taste from anterior 2/3 tongue |
| Posterior auricular nerve | Just after stylomastoid foramen | Motor | Occipitalis, posterior auricular muscle |
| Nerve to stylohyoid & posterior digastric | Just after stylomastoid foramen | Motor | Stylohyoid & posterior belly of digastric |
Localizing Lesions
Because different branches leave the nerve at different levels, the pattern of deficits localizes the lesion:
| Site | Deficits |
|---|---|
| Motor cortex / internal capsule (UMN) | Contralateral lower face weakness only (upper face spared — bilateral cortical supply) |
| Facial nucleus / nerve in pons (LMN) | Ipsilateral entire face weakness |
| Proximal to geniculate ganglion | Entire ipsilateral face palsy + loss of lacrimation + loss of taste anterior 2/3 tongue + hyperacusis |
| Below geniculate ganglion, above chorda tympani | Entire ipsilateral face palsy + loss of taste + hyperacusis; lacrimation intact |
| Stylomastoid foramen (Bell's palsy) | Entire ipsilateral face palsy only; taste, lacrimation, hearing intact (all branches already given off) |
Bell phenomenon: on attempted eye closure with complete palsy, the eye rolls upward — the globe is seen because the eyelid cannot close.
Bell's Palsy
The most common cause of acute facial paralysis (~23/100,000/year). Caused predominantly by HSV-1 reactivation in the geniculate ganglion (HSV-1 DNA isolated by PCR in endoneurial fluid in the majority of surgical cases). Risk factors include diabetes, hypertension, and possibly late pregnancy/postpartum. MRI shows enhancement of the labyrinthine segment and geniculate ganglion with normal HRCT.
Ramsay Hunt syndrome (herpes zoster oticus): VZV reactivation at the geniculate ganglion — facial palsy + ipsilateral ear/auricular vesicles ± hearing loss/vertigo.
Clinical Pearls
- The intimate relationship of CN VII with the parotid gland makes parotidectomy technically demanding; perineural invasion by parotid malignancy can track proximally along the nerve.
- Facial nerve schwannomas present with gradual-onset facial paresis + hearing loss; T1 MRI shows an enhancing, expansile mass along the nerve's course with canal enlargement on HRCT.
- The labyrinthine segment is the narrowest part of the fallopian canal — swelling here (as in Bell's palsy or Ramsay Hunt) readily compresses the nerve.
- The geniculate ganglion is dehiscent (paper-thin overlying bone) in ~25% of ears, a surgical hazard during middle fossa approaches.
Sources: Gray's Anatomy for Students; Adams and Victor's Principles of Neurology, 12th ed.; Cummings Otolaryngology Head and Neck Surgery; Kanski's Clinical Ophthalmology
Mini mental state examination
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
mini mental state examination MMSE scoring sheet
I now have comprehensive content from multiple authoritative textbooks. Here is the full answer:
Mini-Mental State Examination (MMSE)
Overview
The MMSE was developed by Folstein et al. (1975) as a standardized, 30-point bedside screening tool for cognitive function. It remains one of the most widely used mental status examinations across clinical and research settings — community samples, hospitals, specialty clinics — and across a broad range of neurological and psychiatric conditions including dementia, stroke, and depression. Administration typically takes less than 10 minutes and can be performed by trained clinic staff.
Domains Assessed
The MMSE evaluates 11 items across the following cognitive domains:
| Domain | Tasks | Max Points |
|---|---|---|
| Orientation to Time | Year, season, month, date, day | 5 |
| Orientation to Place | Country, county/state, town, hospital, ward/floor | 5 |
| Registration | Name 3 objects; patient repeats them | 3 |
| Attention & Calculation | Serial 7s (subtract 7 from 100, five times) or spell "WORLD" backwards | 5 |
| Recall | Recall the 3 objects from Registration | 3 |
| Language — Naming | Name a pencil and a watch | 2 |
| Language — Repetition | Repeat "No ifs, ands, or buts" | 1 |
| Language — 3-Stage Command | Take paper in right hand, fold it, put it on the floor | 3 |
| Reading | Read and obey "Close your eyes" | 1 |
| Writing | Write a sentence spontaneously | 1 |
| Construction (Visuospatial) | Copy intersecting pentagons | 1 |
| TOTAL | 30 |
Scoring and Interpretation
| Score | Interpretation |
|---|---|
| 27–30 | Normal (no cognitive impairment) |
| 24–26 | Borderline / mild cognitive impairment |
| 18–23 | Mild dementia |
| 10–17 | Moderate dementia |
| < 10 | Severe dementia |
Key threshold: Scores below 24 are generally considered "impaired," though this cutoff must be adjusted for age and education.
Effect of Demographic Variables
Age and education systematically influence MMSE scores, and this is a critical limitation:
- Highly educated patients with early Alzheimer disease may score in the normal range (false negatives)
- Low-education individuals may score in the impaired range without true cognitive impairment (false positives)
Normative data from the Epidemiologic Catchment Area study show, for example, that the mean MMSE score for adults with 0–4 years of education is ~22, while those with 9+ years of education average ~29 — a difference of ~7 points on the same 30-point scale.
Cutoff scores should always be adjusted for age and education before concluding impairment is present.
Uses
- Screening for cognitive impairment (dementia, delirium, stroke effects)
- Serial monitoring — tracking disease progression or treatment response over time
- Severity staging of dementia
- Communication — widely adopted so clinicians share an intuitive sense of what a given score means functionally
The MMSE is not used to make a formal diagnosis, and it does not replace comprehensive neuropsychological testing.
Limitations
- No executive function assessment — other than serial sevens, frontal/executive deficits are not captured; this is a major gap since executive dysfunction is a hallmark of vascular dementia and early Alzheimer disease
- Insensitive to mild cognitive impairment (MCI) — particularly in educated individuals
- Insensitive to noncortical dementias (e.g., Parkinson's dementia, vascular dementia)
- Education and age bias (as above)
- Copyright — the MMSE is a copyrighted instrument (PAR Inc.) requiring purchase for legal clinical or research use
Modified MMSE (3MS)
Developed by Teng and Chui (1987) to address MMSE shortcomings. Key differences:
- 14 items (vs. 11)
- Adds orientation to self, verbal fluency, recognition memory, and executive function (similarities)
- 100-point scoring range (vs. 30) — more sensitive to detecting change
- Impairment cutoff: ≤ 77
- A decline of ≥ 5 points over 5–10 years indicates clinically meaningful cognitive decline
- Age- and education-corrected norms available; regression-based prediction formulas allow more accurate change detection
Comparison with MoCA
| Feature | MMSE | MoCA |
|---|---|---|
| Score range | 0–30 | 0–30 |
| Time | < 10 min | < 10 min |
| Executive function | Minimal | Yes (trail-making, abstraction, clock drawing) |
| Visuospatial | Pentagons only | Clock, cube |
| Target population | Moderate-severe dementia | MCI detection |
| Impairment cutoff | < 24 | < 26 |
| Sensitivity for MCI | Low | 90% |
| Sensitivity for AD | Moderate | 100% |
| Cost | Copyrighted (purchase required) | Free (certification now required in most settings) |
The MoCA (Nasreddine et al., 2005) was designed specifically to overcome the MMSE's insensitivity to MCI and adds probing of executive functioning, working memory, and visuospatial abilities — making it the preferred screening tool when early or subtle cognitive impairment is suspected.
Sources: Bradley and Daroff's Neurology in Clinical Practice; Kaplan & Sadock's Comprehensive Textbook of Psychiatry; Kaplan and Sadock's Synopsis of Psychiatry
Bundle branch block ecg
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching Images
right bundle branch block left bundle branch block ECG pattern comparison
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Loading Image
Excellent — I have comprehensive content and high-quality ECG images from Harrison's and Goldman-Cecil. Here is the full answer:
Bundle Branch Block — ECG
Anatomy & Mechanism
The His-Purkinje system divides into:
- Right bundle branch (RBB) → depolarizes the right ventricle
- Left bundle branch (LBB) → splits into anterior fascicle (left anterior), posterior fascicle (left posterior), and a septal fascicle
Normal ventricular activation: the impulse travels simultaneously down both bundles, depolarizing both ventricles in ~80–100 ms (narrow QRS).
When a bundle branch is blocked, the affected ventricle is depolarized late via slow cell-to-cell conduction rather than rapid Purkinje conduction → wide QRS ≥ 120 ms and abnormal morphology.
General ECG Principles in BBB
- QRS duration ≥ 120 ms (complete BBB) or 110–119 ms (incomplete)
- Secondary ST-T changes — T wave is always opposite in polarity to the last deflection of the QRS (discordance). This is a normal consequence of altered depolarization sequence — not a primary repolarization abnormality
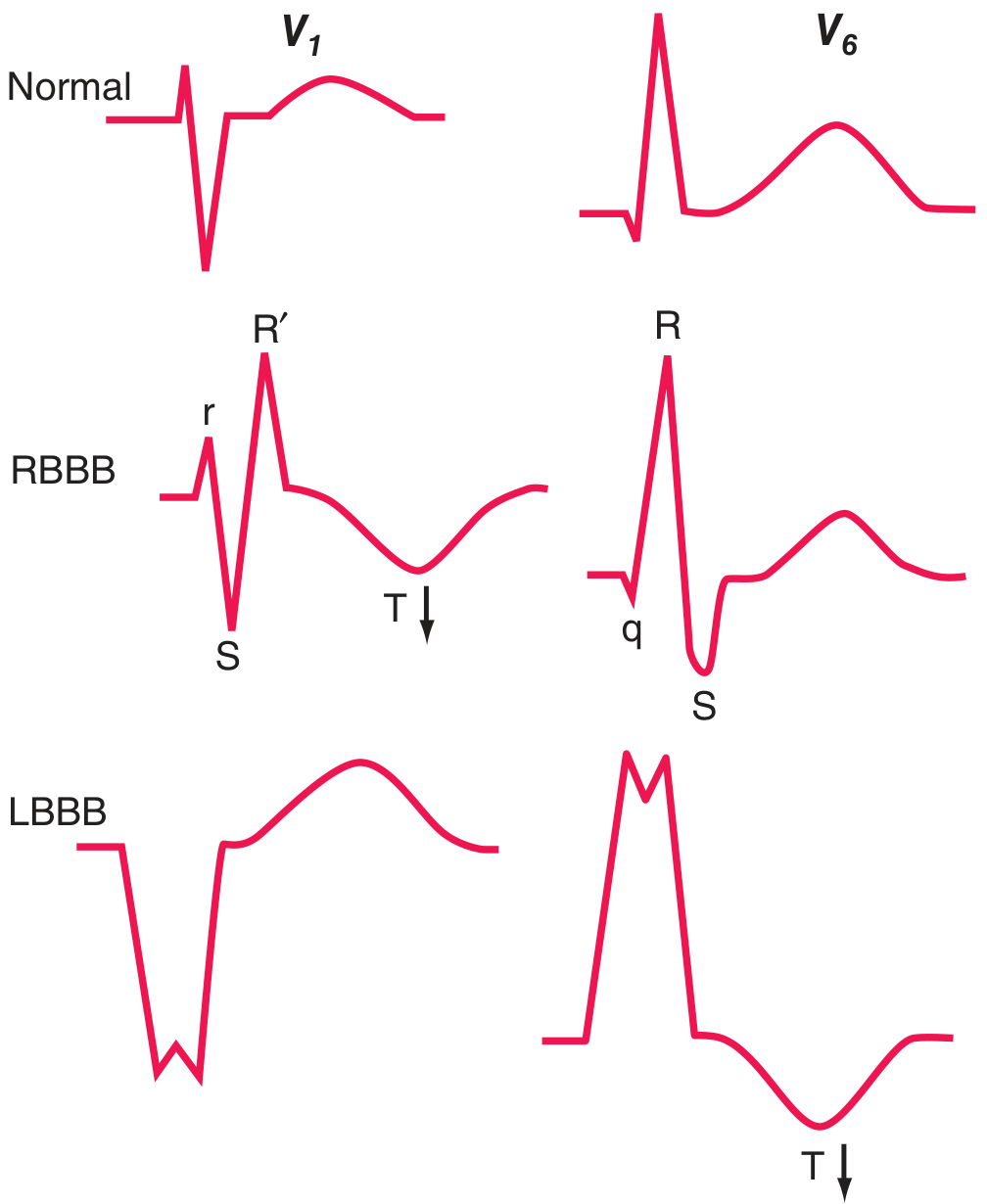
- The "William Morrow" mnemonic: WiLLiaM (LBBB: W in V1, M in V6) / MaRRoW (RBBB: M in V1, W in V6) — see morphology below
Right Bundle Branch Block (RBBB)
ECG Criteria (complete RBBB)
| Feature | Finding |
|---|---|
| QRS duration | ≥ 120 ms |
| V1 morphology | rSR' (or rsR', RSR') — classic "rabbit ears" |
| V5–V6 morphology | Wide, slurred terminal S wave (qRS) |
| Lead I / aVL | Wide, slurred S wave |
| T-wave | Inverted/negative in V1–V2 (secondary change — discordant with terminal R') |
| Axis | Usually normal; septal Q waves (normal septal activation preserved) |
Mechanism
- Left ventricle activates normally (first deflection — small r in V1 from normal septal activation)
- Right ventricle activates late, via spread from the left → terminal vector directed rightward and anteriorly → R' in V1, slurred S in lateral leads
Incomplete RBBB
- QRS 110–119 ms with RSR' in V1–V2, but R' is not taller than the S wave amplitude — often a normal variant, especially in young people and athletes
Left Bundle Branch Block (LBBB)
ECG Criteria (complete LBBB)
| Feature | Finding |
|---|---|
| QRS duration | ≥ 120 ms |
| V1 morphology | Broad rS or QS (dominant deep S, often with notching) |
| V5–V6 / I / aVL | Broad, tall, often notched R wave (no Q wave) — "M-shaped" |
| Septal Q waves | Absent in I, aVL, V5–V6 (septal activation reversed) |
| T-wave | Inverted in I, aVL, V5–V6; upright in V1–V2 (discordant) |
| Axis | Normal or leftward |
Mechanism
- Septal activation reverses: septum activates right-to-left → no initial q wave in lateral leads
- Left ventricle depolarizes late via slow spread from right → broad, notched R waves in lateral leads and a wide QS/rS in V1
Waveform Comparison — Normal vs. RBBB vs. LBBB
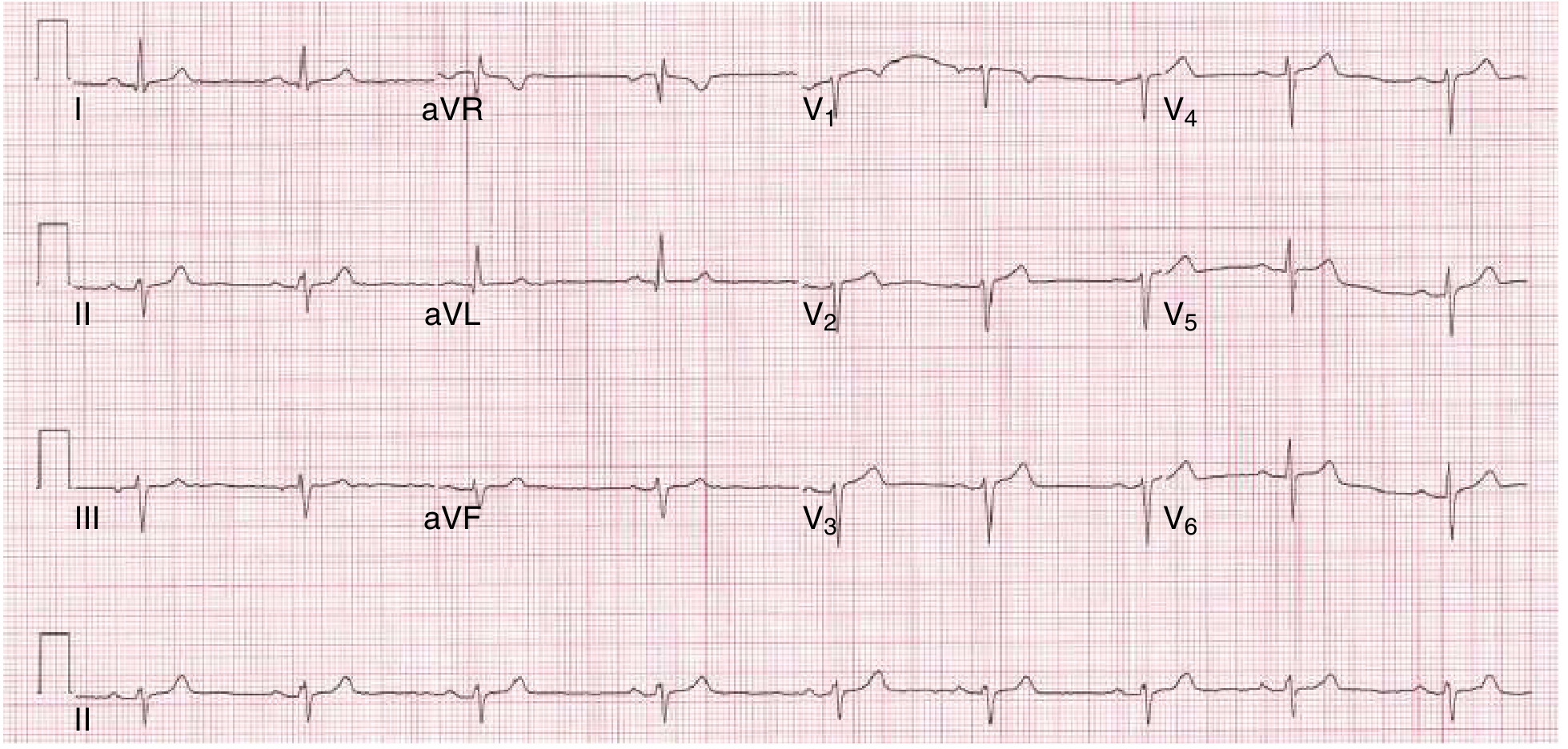
12-Lead ECG Examples
RBBB (left anterior fascicular block + RBBB = bifascicular block):
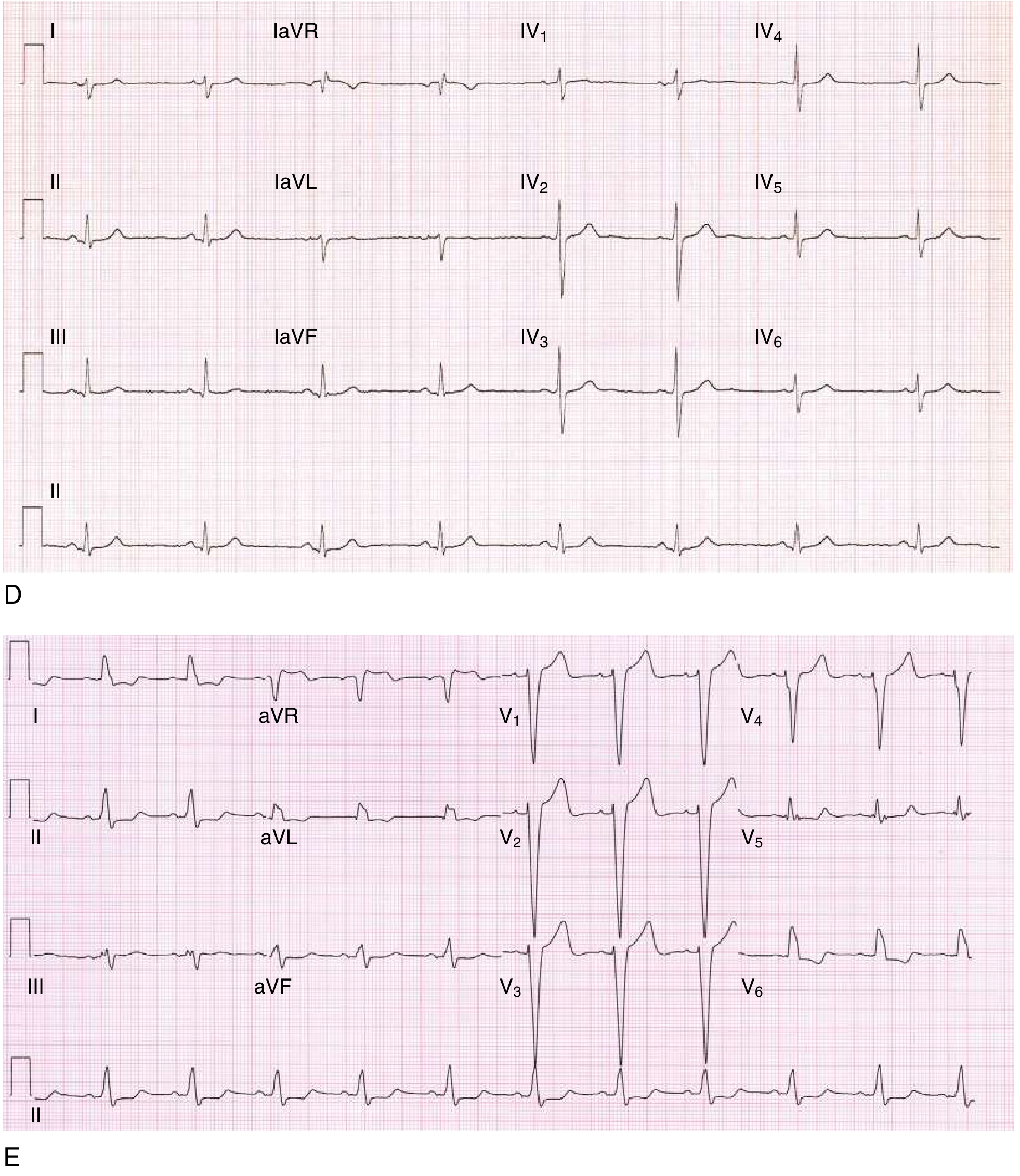
Left posterior fascicular block and LBBB:
Fascicular Blocks (Hemiblocks)
These block the left bundle's sub-fascicles. QRS is not significantly widened (<120 ms) but the frontal axis shifts:
| Block | Axis | V leads | Features |
|---|---|---|---|
| Left Anterior Fascicular Block (LAFB) | ≤ −45° (marked LAD) | Late precordial transition | Small q in I, aVL; small r in II, III, aVF |
| Left Posterior Fascicular Block (LPFB) | ≥ +110° (RAD) | Delayed R progression | Small r in I, aVL; q in II, III, aVF — rare; must exclude other causes of RAD |
Bifascicular & Trifascicular Block
| Pattern | Components | Risk |
|---|---|---|
| Bifascicular block | RBBB + LAFB (most common) or RBBB + LPFB | ~6%/year risk of complete AV block |
| Trifascicular block | Bifascicular + long PR interval (1st-degree AV block) | High risk for complete AV block |
| Alternating BBB | RBBB on some beats, LBBB on others | Class I indication for permanent pacing |
BBB in Acute MI
LBBB alters ST-T morphology throughout the precordium, masking ST-elevation MI. Use Sgarbossa criteria to identify MI in LBBB:
| Criterion | Points |
|---|---|
| ST elevation ≥ 1 mm concordant with QRS (same direction) | 5 |
| ST depression ≥ 1 mm in V1, V2, or V3 (concordant with negative QRS) | 3 |
| ST elevation ≥ 5 mm discordant with QRS (opposite direction — normally expected but excessively large) | 2 |
Score ≥ 3 = likely acute MI. New LBBB in the setting of chest pain = STEMI equivalent until proven otherwise — treat with emergent reperfusion.
Risk of progression to complete heart block in BBB during MI:
- RBBB + LAFB → 27%; RBBB + LPFB → 29%; alternating BBB → 44%
Causes
| RBBB | LBBB |
|---|---|
| Can be normal (no structural disease) | Almost always signifies structural disease |
| Atrial septal defect | Coronary artery disease / ischemic cardiomyopathy |
| Pulmonary hypertension / embolism | Hypertensive heart disease |
| Right heart disease | Aortic valve disease / post-TAVR |
| Congenital heart disease | Dilated cardiomyopathy |
| Brugada pattern (RBBB morphology + STE in V1–V3) | Fibrosis of conduction system |
LBBB is often a marker of four conditions with increased cardiovascular morbidity: CAD, hypertension, aortic valve disease, and cardiomyopathy. LBBB-associated ventricular dyssynchrony worsens heart failure; these patients benefit from cardiac resynchronization therapy (CRT / biventricular pacing).
Rate-Related BBB
BBB may be intermittent, appearing only when the heart rate exceeds a critical threshold — most commonly phase 3 (tachycardia-dependent) aberrancy. This is distinct from fixed (chronic) BBB.
Sources: Harrison's Principles of Internal Medicine 22nd ed.; Goldman-Cecil Medicine; Fuster and Hurst's The Heart, 15th ed.; Roberts and Hedges' Clinical Procedures in Emergency Medicine; Pfenninger and Fowler's Procedures for Primary Care
Syncope
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material across all domains. Here is the complete answer:
Syncope
Definition
Syncope is an abrupt, transient, and complete loss of consciousness with inability to maintain postural tone, followed by spontaneous, complete recovery. The underlying mechanism is always transient cerebral hypoperfusion. Episodes that are incomplete (i.e., lightheadedness without loss of consciousness) are termed presyncope and carry similar clinical significance.
Syncope must be distinguished from cardiac arrest (loss of consciousness without spontaneous recovery) and from seizure, hypoglycemia, and psychogenic pseudo-syncope.
Epidemiology
- Lifetime prevalence: ~40%; recurrence rate: 14%
- Accounts for 1–3% of ED visits and 6% of hospital admissions
- Annual cost of syncope hospitalizations:
$1.7 billion ($26,000/hospitalization) - Incidence increases with age; in the Framingham Study: 3.0% in men, 3.5% in women over 26 years
- Cause remains unknown in up to 50% of patients despite extensive evaluation
Pathophysiology
The common final pathway is a fall in cerebral perfusion below the threshold for consciousness. This can result from:
- Reduced cardiac output (arrhythmia, obstruction)
- Reduced systemic vascular resistance (vasodilation)
- Reduced venous return / preload (hypovolemia, venous pooling)
- Combinations of the above
Classification & Causes
1. Reflex (Neurally Mediated) Syncope — Most Common
Caused by inappropriate vasodilation, bradycardia, or both due to sudden failure of autonomic tone.
| Type | Mechanism / Trigger | Features |
|---|---|---|
| Vasovagal (neurocardiogenic) | Prolonged standing, emotional stress, pain, venipuncture, warm/crowded environment | Prodrome: nausea, pallor, diaphoresis, lightheadedness, warmth, blurred vision; more common in young people |
| Carotid sinus syndrome | Pressure on carotid sinus (shaving, tight collar, head turning) → sinus arrest/AV block (cardioinhibitory) and/or vasodilation (vasodepressor) | More common in men ≥50 years; positive carotid sinus massage: pause ≥3 s and/or SBP drop ≥50 mmHg |
| Situational | Micturition, defecation, coughing, swallowing, laughing, sneezing, post-exercise, postprandial | Closely linked to a specific provoking action |
Mechanism of vasovagal syncope: venous pooling in dependent areas → decreased venous return → reflex increase in myocardial contractility → activation of ventricular mechanoreceptors (normally fired only during stretch) → paradoxic decrease in sympathetic outflow (hypotension) + increased vagal tone (bradycardia → sometimes asystole on tilt-table).
2. Orthostatic Syncope
Defined as a fall in SBP ≥20 mmHg and/or DBP ≥10 mmHg within 3 minutes of standing.
| Subtype | Examples |
|---|---|
| Hypovolemia | Dehydration, hemorrhage, Addison disease |
| Drug-induced | Antihypertensives (β-blockers, CCBs, nitrates, ACEi/ARBs, diuretics), antidepressants (TCAs, MAOIs, SSRIs), antipsychotics, levodopa, opioids, alcohol |
| Autonomic dysfunction | Diabetes, Parkinson disease, multisystem atrophy, amyloidosis |
| Deconditioning / prolonged bed rest |
3. Cardiac Syncope — Highest Mortality
Cardiac syncope from cardiovascular disorders is associated with ~50% mortality over 5 years and ~30% in the first year. Must be actively excluded.
Arrhythmic causes:
- Sinus node dysfunction (sinus bradycardia < 40 bpm, sinus pauses > 3 s)
- AV block (Mobitz II, complete heart block)
- Supraventricular tachycardias (SVT, AF/flutter with rapid ventricular response)
- Ventricular tachycardia / fibrillation
- Long QT syndrome (congenital or acquired), Brugada syndrome, CPVT
- Pacemaker dysfunction
Structural / obstructive causes:
- Aortic stenosis (syncope = severe obstruction, poor prognosis)
- Hypertrophic cardiomyopathy (exertional syncope = high SCD risk)
- Mitral stenosis
- Atrial myxoma
- Prosthetic valve dysfunction
- Pulmonary embolism / pulmonary hypertension
- Cardiac tamponade
- Acute MI / ischemia
4. Cerebrovascular
- Vertebrobasilar TIA / insufficiency — syncope nearly always preceded by vertigo, diplopia, dysarthria, ataxia
- Subclavian steal syndrome — upper extremity exertion shunts blood retrograde via vertebral artery
- Occlusive disease (Takayasu arteritis, aortic arch syndrome)
- Klippel-Feil deformity / cervical spondylosis (vertebral artery compression)
Syncope vs. Seizure — Key Differentiators
| Feature | Syncope | Seizure |
|---|---|---|
| Posture-related | Common | No |
| Skin color | Pallor | Cyanosis or normal |
| Diaphoresis | Common | Rare |
| Prodrome | Often prolonged (minutes) | Brief aura |
| Convulsions | Rare; minor irregular twitching | Common; rhythmic jerks |
| Tongue biting | No | Common |
| Urinary incontinence | Rare | Common |
| Postictal confusion | Rare | Common |
| Postictal headache | No | Common |
| ECG/EEG | Normal | Often abnormal |
Syncope has been misdiagnosed as seizure in 38% of patients who continued to have episodes despite anticonvulsant therapy — a critical clinical trap.
Clinical Approach
History
Key clinical context questions:
- Position at onset — lying/sitting/standing (orthostatic vs. cardiac)?
- Activity — exertional (cardiac outflow obstruction, arrhythmia, ARVC, CPVT) vs. at rest?
- Prodrome — nausea/diaphoresis (vasovagal) vs. palpitations (arrhythmia) vs. none (cardiac, elderly)?
- Duration — brief and sudden = cardiac; gradual onset with prodrome = vasovagal
- Recovery — postictal confusion? (seizure); rapid and complete = syncope
- Triggers — specific situational triggers, head turning (carotid sinus), venipuncture, crowds
- Medications — QT-prolonging drugs, antihypertensives, antiparkinsonian agents
- Family history — sudden cardiac death (long QT, HCM, Brugada, ARVC)
Physical Examination
- Orthostatic BP (only useful if symptoms reproduced on standing)
- Blood pressure in both arms (subclavian steal, Takayasu)
- Heart rate during arrhythmia: >140 bpm = likely not sinus; <40 bpm = likely complete AV block
- Cardiac auscultation — murmurs (aortic stenosis: systolic ejection murmur; HCM: dynamic murmur)
- Signs of heart failure; new neurological deficit (not syncope — consider stroke/SAH)
- Carotid sinus massage (older patients, not routine in ED)
Investigations
ECG (12-lead) — Recommended in nearly all cases
ECG findings diagnostic of arrhythmic syncope:
- Sinus bradycardia < 40 bpm or pause > 3 s
- Mobitz II second-degree AV block
- Complete (third-degree) heart block
- Sustained SVT or VT
ECG findings raising concern for underlying cause:
- Prolonged QTc → long QT syndrome
- Short PR + delta wave → WPW (pre-excitation)
- RBBB + ST elevation V1–V3 → Brugada syndrome
- RV strain pattern → PE
- New LBBB → rule out STEMI
- Epsilon waves / RBBB + T-wave inversions V1–V3 → ARVC
Additional Testing
| Test | When |
|---|---|
| Holter monitor (24–48 h) | Suspected arrhythmia, frequent episodes |
| Event recorder (weeks) | Infrequent episodes |
| Implantable loop recorder (ILR) | Very infrequent episodes (weeks–months); diagnostic in ~2/3 of unexplained syncope |
| Echocardiogram | Suspected structural heart disease |
| Cardiac MRI | ARVC, HCM, inconclusive echo |
| Exercise stress test | Exertional syncope |
| Tilt-table test | Suspected neurocardiogenic or orthostatic syncope |
| Electrophysiology study (EPS) | HV interval ≥70 ms or infranodal block → pacing indication |
| Troponin | Only if ACS is suspected |
| BNP/NT-proBNP | Prognostic utility for 30-day adverse cardiac events |
| CT head | Only if head trauma after syncope, or new neurological deficit |
Routine labs (CBC, electrolytes, glucose) are not supported by evidence for standard syncope evaluation.
Prognosis
| Cause | Mortality |
|---|---|
| Cardiac syncope | ~50% at 5 years; ~30% in first year; all-cause mortality doubled vs. general population |
| Non-cardiac (reflex/orthostatic) | ~30% at 5 years; <10% in first year; more benign course |
| Unexplained syncope | 6–10% at 3 years; 24% at 5 years |
| Vasovagal / reflex | Benign; recurrence affects quality of life |
Syncope is an ominous sign in HCM (high SCD risk) and in aortic stenosis (severe obstruction, < 2–3 year survival without intervention).
Management
Reflex (Vasovagal) Syncope
- Non-pharmacologic first: education, avoid triggers, physical counterpressure maneuvers (leg crossing, arm tensing), liberal salt and fluid intake, compression stockings, tilt training
- Midodrine (α-agonist) — FDA-approved for orthostatic hypotension; reduces vasovagal recurrence
- SSRIs — randomized controlled trial evidence for prevention of recurrent neurocardiogenic syncope
- Fludrocortisone — volume expansion; evidence mixed
- Ivabradine — useful in POTS and neurocardiogenic syncope refractory to β-blockers
- Pyridostigmine — acetylcholinesterase inhibitor; prevents orthostatic BP falls without worsening supine hypertension
- Cardiac pacing — only in cardioinhibitory carotid sinus syndrome with documented pause; not routinely for vasovagal
Orthostatic Syncope
- Treat underlying cause; review/stop offending medications
- Volume expansion (fluids, salt, fludrocortisone)
- Midodrine, droxidopa (approved for neurogenic orthostatic hypotension)
- Head-of-bed elevation (reduces nocturnal natriuresis)
Cardiac Syncope
- Arrhythmic: permanent pacemaker (bradycardia, heart block); ICD (VT/VF, Brugada, long QT, HCM); catheter ablation (SVT, VT); antiarrhythmic drugs
- Structural: treat underlying disease (valve surgery/TAVR for AS; septal reduction for HCM; anticoagulation for PE)
- Bifascicular block with syncope + HV ≥70 ms on EPS: permanent pacing (Class I indication)
- Alternating BBB: pacemaker regardless of symptoms (Class I)
Sources: Fuster and Hurst's The Heart, 15th ed.; Bradley and Daroff's Neurology in Clinical Practice; Rosen's Emergency Medicine; Tintinalli's Emergency Medicine
This is a shared conversation. Sign in to Orris to start your own chat.