What are the various causes of gynecomastia? And the treatment for each
Reading File
Finding Sources
Searching PubMed
gynecomastia causes treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive information to compile a thorough answer. Here is the complete overview:
Gynecomastia: Causes and Treatment
Gynecomastia is benign enlargement of the male breast glandular tissue (>2–4 cm diameter), driven by an increased estrogen/androgen ratio. It must be distinguished from pseudogynecomastia (lipomastia — fat accumulation without glandular proliferation). The underlying mechanism in virtually every cause is either excess estrogen, androgen deficiency, or disrupted androgen action.
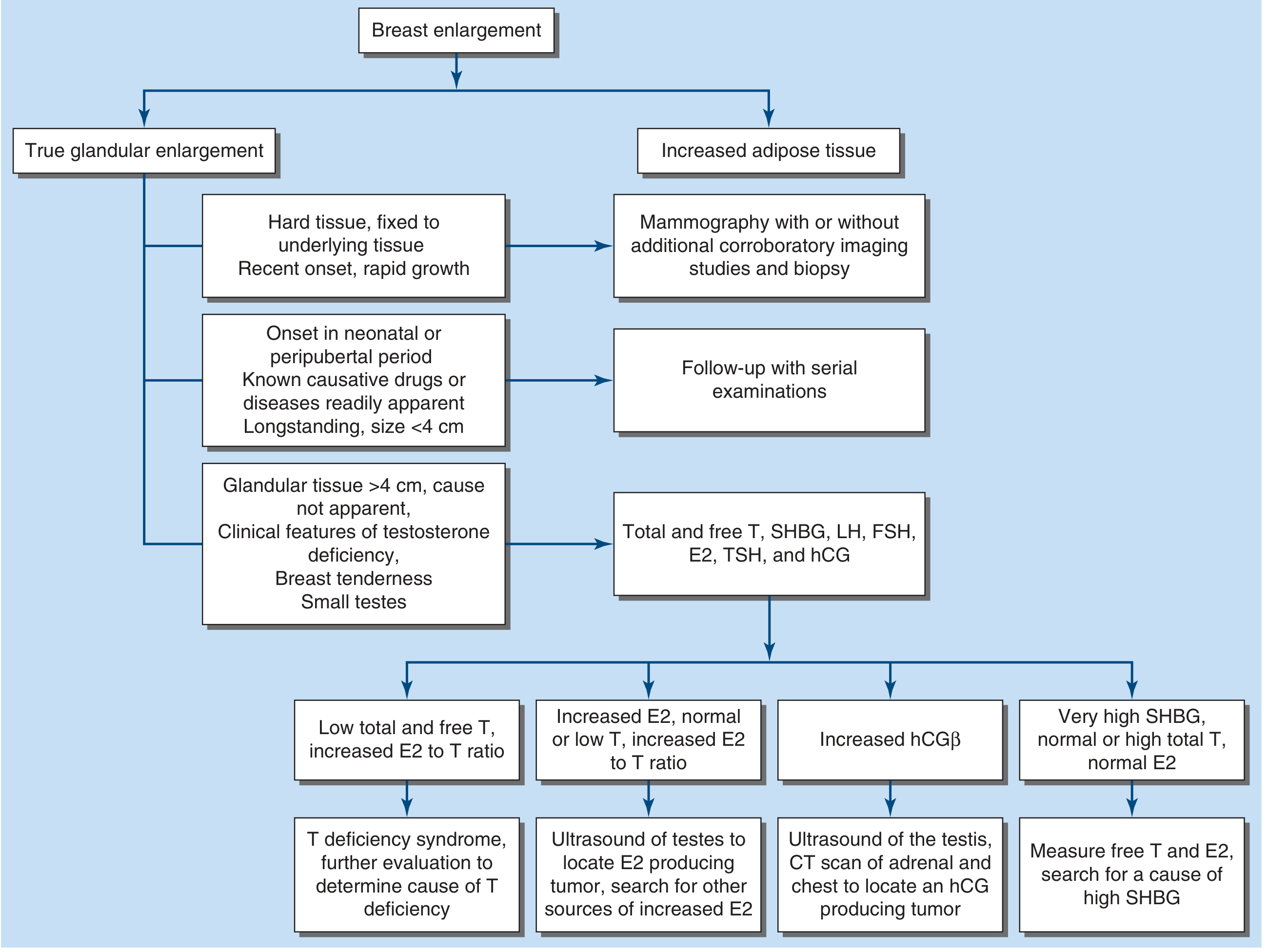
I. Physiologic (Normal) Causes
These three life stages produce gynecomastia transiently and require no treatment beyond reassurance.
| Phase | Mechanism | Treatment |
|---|---|---|
| Neonatal | Transplacental transfer of maternal/placental estrogens | Resolves spontaneously within weeks |
| Pubertal (ages 12–15; 40–70% of boys) | Early puberty has a high estradiol-to-testosterone ratio | Reassurance; resolves by age 18 in most cases |
| Senescent (men >50–70 yrs) | Falling testosterone, rising SHBG, increased adipose aromatase activity → relative hyperestrism | Usually bilateral; treat underlying contributors (weight loss, medication review) |
II. Estrogen Excess States
A. Tumors Secreting Estrogen or hCG
| Tumor | Mechanism | Treatment |
|---|---|---|
| Leydig cell tumor of testis | Direct estradiol secretion | Surgical excision (orchiectomy or testis-sparing) |
| Sertoli cell tumor (including Peutz-Jeghers syndrome, Carney complex associations) | Direct estrogen or aromatase excess; Sertoli tumors in Peutz-Jeghers aromatize androgens to estrogens; prepubertal onset with premature epiphyseal closure | Excision; treat the syndrome (surveillance for hamartomas, pigmentation) |
| Granulosa-theca cell tumor of testis | Estrogen secretion | Surgical excision |
| Germ cell tumors (choriocarcinoma, seminoma, teratoma, embryonal carcinoma) | hCG secretion → stimulates Leydig cell estrogen synthesis | Orchiectomy ± chemotherapy/radiation per tumor type |
| Adrenal cortical neoplasm | Secrete estrogen precursors (androstenedione) → peripheral aromatization; or direct estrogen | Adrenalectomy |
| Hepatocellular carcinoma | Can produce estrogens; also liver disease reduces estrogen catabolism | Resection/transplant if feasible; oncologic management |
| Lung carcinoma | Ectopic hCG production | Oncologic management |
| True hermaphroditism | Both ovarian and testicular tissue present | Surgical gonadal management tailored to karyotype/phenotype |
B. Endocrine Disorders
| Disorder | Mechanism | Treatment |
|---|---|---|
| Hyperthyroidism | Elevated SHBG raises the free E2/free T ratio; also increased conversion of androstenedione to estrogen | Antithyroid therapy (methimazole, propylthiouracil), radioiodine, or thyroidectomy → gynecomastia often resolves |
| Hypothyroidism | Altered sex hormone metabolism | Thyroid hormone replacement |
| Congenital adrenal hyperplasia (CAH) | Increased availability of androstenedione substrate for aromatization | Glucocorticoid suppression of adrenal androgen overproduction |
C. Liver Disease
Alcoholic and non-alcoholic cirrhosis: Reduced hepatic catabolism of androstenedione → shunting to peripheral aromatase sites; also alcohol directly inhibits testicular testosterone synthesis and increases SHBG.
Treatment: Abstinence from alcohol, management of liver disease; gynecomastia may partially regress.
D. Nutritional/Refeeding Gynecomastia
After starvation (e.g., POW camps historically, anorexia recovery), resumption of nutrition restores pituitary gonadotropin secretion after a period of shutdown, causing a transient estrogen surge.
Treatment: Nutritional rehabilitation; resolves spontaneously.
E. Familial Aromatase Excess Syndrome
A rare autosomal dominant or X-linked disorder caused by CYP19 mutation or chromosomal rearrangement (15q21 inversion creating chimeric CYP19), leading to excessive estrogen production in fat and extragonadal tissues. Features include prepubertal/peripubertal gynecomastia, advanced bone age, short stature (premature epiphyseal closure), and hypogonadotropic hypogonadism.
Treatment: Aromatase inhibitors (anastrozole, letrozole) — most effective in the early proliferative phase; surgery for established fibrotic disease.
F. Obesity
Increased adipose mass → increased aromatase activity → increased peripheral conversion of androgens to estrogens.
Treatment: Weight loss is first-line; reduces aromatase substrate. Pharmacologic or surgical options if persistent.
III. Androgen Deficiency States (Hypogonadism)
When testosterone falls, residual adrenal and gonadal androgens are still aromatized to estrogens → relative hyperestrism.
A. Primary Testicular Failure (Hypergonadotropic Hypogonadism)
| Condition | Notes | Treatment |
|---|---|---|
| Klinefelter syndrome (47,XXY) | Most common; hypergonadotropic hypogonadism + azoospermia; also carries increased male breast cancer risk | Testosterone replacement therapy (TRT); surgery for persistent gynecomastia |
| Reifenstein syndrome | Partial androgen insensitivity | TRT may not help (due to androgen resistance); surgical mastectomy |
| Androgen insensitivity disorders | AR mutations → androgens cannot act; estrogens go unopposed | Usually managed surgically |
| Kallmann syndrome | GnRH deficiency + anosmia | GnRH pulsatile therapy or gonadotropin (hCG/FSH) therapy to restore testosterone |
| Kennedy's disease (spinobulbar muscular atrophy) | CAG repeat expansion in AR gene → androgen resistance | Supportive; TRT partially beneficial |
| Congenital anorchia / eunuchoidal state | No testicular tissue → no testosterone | TRT |
| Hereditary defects in androgen biosynthesis | Enzyme deficiencies (e.g., 17β-HSD, 17α-hydroxylase) | TRT; corticosteroids if CAH overlap |
| Orchitis (e.g., mumps), trauma, irradiation, cryptorchidism | Direct testicular damage | TRT if testosterone deficient; treat underlying cause |
B. Secondary Testicular Failure (Hypogonadotropic Hypogonadism)
Causes include pituitary tumors, hyperprolactinemia, hemochromatosis, and ACTH deficiency.
Treatment: Directed at the underlying pituitary/hypothalamic cause (e.g., dopamine agonists for prolactinoma, testosterone supplementation).
C. Androgen Deprivation Therapy (ADT) for Prostate Cancer
GnRH agonists (leuprolide, goserelin) ± androgen receptor blockers (enzalutamide, bicalutamide) are a very common iatrogenic cause of painful gynecomastia in men with prostate cancer.
Treatment:
- Tamoxifen 20 mg/day is the most effective option — both preventive and therapeutic; reduces breast pain and size in >50% of patients
- Radiotherapy (prophylactic low-dose breast irradiation) before ADT initiation can prevent gynecomastia
- Aromatase inhibitors: less effective than tamoxifen for ADT-associated gynecomastia
- Surgery if medical therapy fails
D. Renal Failure
Regardless of etiology, chronic renal failure causes gynecomastia through multiple mechanisms (increased LH, decreased testosterone, increased prolactin).
Treatment: Dialysis may partially improve; testosterone replacement considered; surgery if persistent.
IV. Drug-Induced (Pharmacologic) Causes
Drugs are one of the most common identifiable causes. Mechanisms:
| Mechanism | Examples |
|---|---|
| Direct estrogenic activity | Exogenous estrogens, oral contraceptive pills, digitalis (digoxin), phytoestrogens, anabolic steroids, marijuana |
| Stimulate estrogen synthesis | hCG, clomiphene |
| Inhibit androgen synthesis | GnRH agonists/antagonists, ketoconazole |
| Block androgen receptor | Spironolactone, flutamide, bicalutamide, enzalutamide, cyproterone acetate |
| Unknown/idiopathic mechanism | Cimetidine, imatinib, antiretroviral drugs (HIV therapy — especially efavirenz, stavudine), reserpine, theophylline, verapamil, tricyclic antidepressants, furosemide, diazepam, phenytoin |
Treatment: Discontinue or substitute the offending drug — this is the single most important step and often leads to resolution over weeks to months. If the drug cannot be stopped (e.g., ADT for prostate cancer), add tamoxifen.
V. Idiopathic
In a significant proportion of patients (~25%), no cause is found despite thorough evaluation. May represent subtle perturbations in the estrogen/androgen ratio.
Treatment: Watchful waiting; anti-estrogen therapy if symptomatic.
Summary of Treatment Strategies
| Treatment | Indication | Notes |
|---|---|---|
| Remove/correct the cause | All cases where feasible | First-line; most effective if done early |
| Testosterone replacement (TRT) | Hypogonadism (primary/secondary) | Effective when androgen deficiency is the cause |
| Tamoxifen 20 mg/day | Painful/progressive gynecomastia; ADT-associated; medical therapy | Reduces pain and size in >50%; drug of choice for most medical management |
| Raloxifene | Alternative SERM | Similar to tamoxifen; some evidence in pubertal gynecomastia |
| Aromatase inhibitors (anastrozole, letrozole) | Early proliferative-phase gynecomastia; aromatase excess syndrome | Anastrozole was not superior to placebo in established (fibrotic) gynecomastia in RCT |
| Danazol | Refractory cases | Works, but significant androgenic side effects limit use |
| Surgery (subcutaneous mastectomy/liposuction) | Long-standing gynecomastia (>1 year → fibrosis); severe distress; failed medical therapy; suspected malignancy | Most effective for established disease; grading guides technique |
| Prophylactic breast irradiation | Prevention in men starting ADT | Given before ADT to prevent breast enlargement |
Key principle: Gynecomastia present for >1 year typically undergoes fibrous replacement and will not respond to medications — surgery is then necessary for symptom relief.
Sources: Harrison's Principles of Internal Medicine 22e (2025), pp. 3159–3160; Schwartz's Principles of Surgery 11e, pp. 576–577; Textbook of Family Medicine 9e, p. 1045
This is a shared conversation. Sign in to Orris to start your own chat.